How is hazardous waste treated using the electrolysis process?
There are several solutions for hazardous-waste handling. The most ideal option is to decrease waste at its source, or to recycle the resources for another useful purpose. Nonetheless, while reduction and recycling are attractive possibilities, they are not seen as the final solution, to the hazardous-waste disposal problem.
There will always be a need for processes like electrolysis,for hazardous waste treatment.
What is hazardous waste?
Hazardous waste is defined as waste that has qualities that makes it potentially hazardous, to human health or the environment. Hazardous waste might take the form of liquids, solids, or gases. They might be manufacturing by-products, discarded used materials, or abandoned unused commercial items, like cleaning fluids (solvents) or insecticides, etc.
List of hazardous waste
Wastes may be classified as hazardous waste if they display any of the four hazardous waste criteria, i.e., ignitability, corrosivity, reactivity, and toxicity. Here are some examples of hazardous waste:
· Solvent wastes from many major manufacturing and industrial processes,
· Manufacturing of pesticides or petroleum refining,
· Commercial chemical items,
· Chemicals used in industry, insecticides, and pharmaceuticals,
· Mercury-containing products that are discarded, etc.
What is electrolysis?
The technique of creating a chemical reaction by passing an electric current, through a substance or mixture of chemicals, usually in liquid form, is known as electrolysis. As a result, electricity is defined as the movement of electrons or ions. Ions must be present in the compound for electrolysis to function. Thus, it is the process of breaking down the materials. It frequently decomposes the chemical into its constituents.
Can electrolysis be helpful to treat hazardous waste?
Electrolysis technology is a crucial and sophisticated process in many aspects of wastewater treatment, including clean synthesis, monitoring of pollutant removal efficiency, water sterilization, clean energy conversion, and efficient storage and electricity consumption.
Electrolysis provides several advantages, including simple equipment, ease of operation, and the elimination of the requirement for chemicals, for sedimentation and flock generation. This enables wastewater treatment to be electrochemically oxidized, or to convert organic pollutants to non-hazardous inorganic compounds.
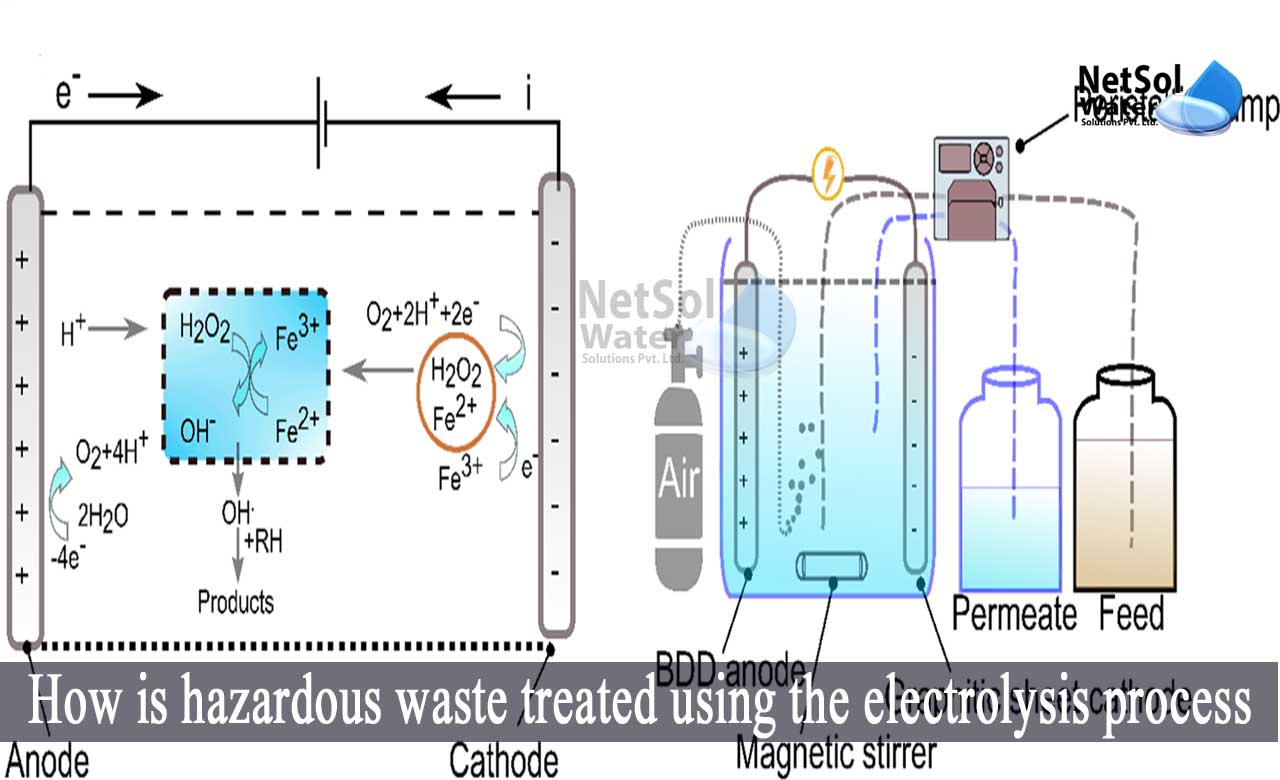
How is hazardous waste treated using electrolysis?
The main process of electrolytic reactions begins at the electrode surface, clot formation in the aqueous phase, absorption of soluble or friction pollutants, and removal via sedimentation and flotation.
When the water container is electrolyzed, hydroxyl and hydronium ions are formed, which flows through the fluid from one electrode to the next. These ions, particularly the hydroxyl ions, will come into contact with bacteria and chemically react with them, to eliminate them.
By eliminating salts from this water, electrolysis allows recycling to occur. The electric field generated by the electrodes provides the driving force. In chlorine water, it eliminates the chloride component of the water.
Advantages of electrolysis
The technique provides a number of benefits, including the following:
1) It is more successful at destabilizing small friction particles.
2) It can do simultaneous freezing and flotation while producing little sludge.
3) The devices are small and compact, making them ideal for placement in tight quarters.
Conclusion
Electrolysis is a reasonably simple process with oxidation or reduction chemistry, with the potential benefits of operating at room temperature and pressure.
Netsol Water offers the most reliable solutions, like wastewater treatment plants, using electrochemical (electrolysis) wastewater treatment technology, for better waste treatment and waste management. Considering its great efficacy, low maintenance cost, minimal labour demand, and speedy effects, electrochemical treatment is an ideal treatment option.
Netsol Water is Greater Noida-based leading water & wastewater treatment plant manufacturer. We are industry's most demanding company based on client review and work quality. We are known as best commercial RO plant manufacturers, industrial RO plant manufacturer, sewage treatment plant manufacturer, Water Softener Plant Manufacturers and effluent treatment plant manufacturers. Apart from this 24x7 customer support is our USP. Call on +91-9650608473, or write us at enquiry@netsolwater.com for any support, inquiry or product-purchase related query.