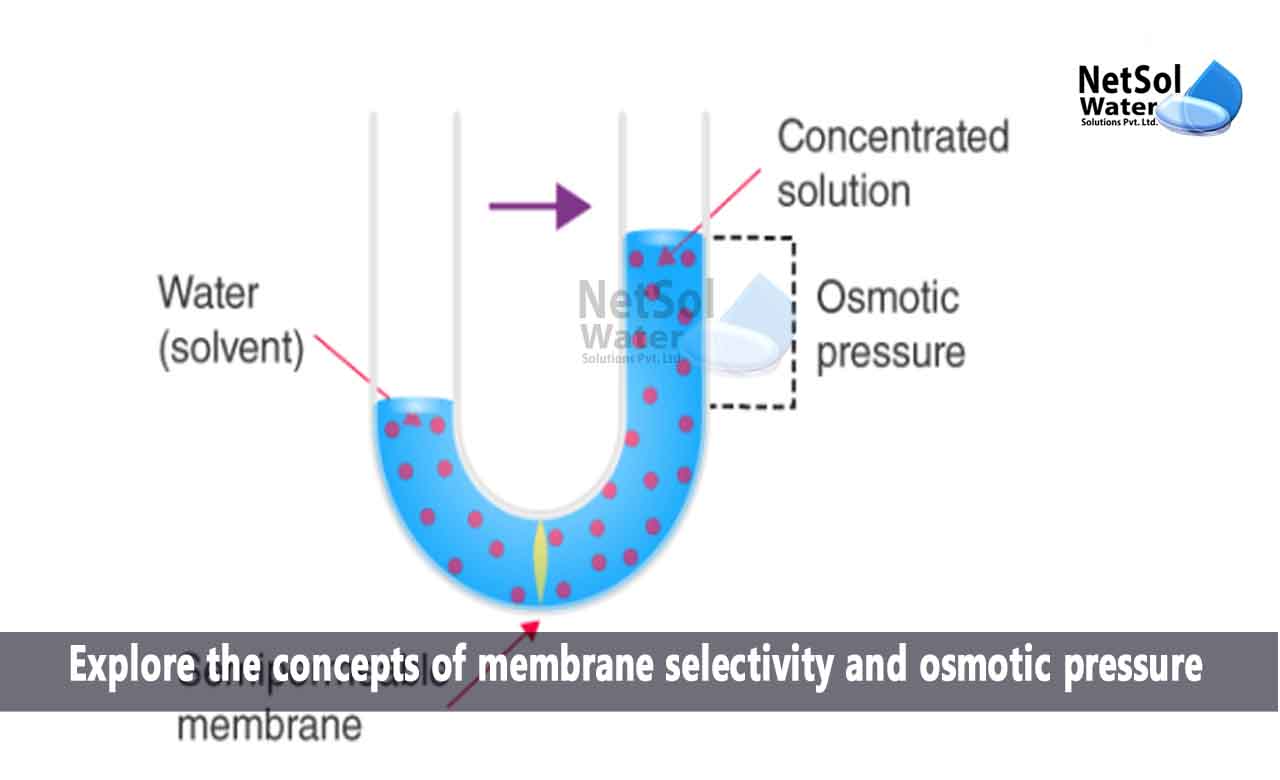
What is the concepts of membrane selectivity and osmotic pressure?
Reverse osmosis (RO) is a powerful water purification technology that relies on the scientific principles of membrane selectivity and osmotic pressure. By understanding the underlying science behind reverse osmosis, we can gain insight into how this process effectively removes contaminants from water, providing clean and purified drinking water. In this blog, we will explore the concepts of membrane selectivity and osmotic pressure, shedding light on the science behind reverse osmosis and its applications in various industries.
Membrane Selectivity:
The foundation of reverse osmosis lies in the selectivity of the semi-permeable membrane. RO membranes are designed to allow the passage of water molecules while selectively blocking the passage of dissolved solutes and contaminants. This selectivity is primarily determinedby the pore size and the chemical properties of the membrane.
RO membranes typically consist of thin film composites (TFC) or cellulose acetate. These materials have densely packed polymer chains that form a matrix of microscopic pores. The size of these pores is crucial for determining the membrane's selectivity.
The pores in an RO membrane are incredibly small, typically ranging from 0.0001 to 0.001 micrometers. This size is significantly smaller than the majority of dissolved solutes and contaminants found in water. As a result, water molecules, which are smaller, can pass through the membrane, while larger particles, such as ions, bacteria, viruses, and even some dissolved organic molecules, are effectively blocked.
In addition to pore size, the chemical properties of the membrane surface play a role in selectivity. Many RO membranes have hydrophilic surfaces, meaning they have an affinity for water molecules. This hydrophilicity helps to attract water and repel hydrophobic solutes, further enhancing the membrane's ability to selectively allow water passage while inhibiting the passage of contaminants.
Osmotic Pressure:
Osmotic pressure is a fundamental concept in reverse osmosis. It is the pressure requiredto counteract the natural osmosis process, where water moves from a region of lower solute concentration to a region of higher solute concentration through a semi-permeable membrane.
In reverse osmosis, external pressure is applied to the more concentrated solution to overcome the natural osmotic pressure. This pressure allows water molecules to move against the concentration gradient, from the more concentrated side to the less concentrated side of the membrane. As a result, contaminants and dissolved solutes are left behind, while purified water passes through the membrane.
The applied pressure must exceed the osmotic pressure to achieve effective reverse osmosis. By doing so, the natural flow of water is reversed, and the process becomes highly efficient in removing impurities and producing clean water.
Conclusion:
Understanding the science behind reverse osmosis is essential for appreciating its efficacy in water purification. Membrane selectivity, determinedby pore size and chemical properties, enables the membrane to selectively allow the passage of water molecules while blocking contaminants. This selective barrier ensures the production of clean and purified water.
Osmotic pressure, on the other hand, is overcome by applying external pressure to the more concentrated solution. This pressure allows the water to move against the concentration gradient, resulting in the separation of contaminants from the purified water.
By comprehending the scientific principles of membrane selectivity and osmotic pressure in reverse osmosis, we can recognize the value of this technology in various industries, including water treatment, desalination, and residential water purification. Reverse osmosis continues to be a reliable and efficient method for obtaining high-quality drinking water, promoting health and well-being for communities worldwide.
Netsol Water is Greater Noida-based leading water & wastewater treatment plant manufacturer. We are industry's most demanding company based on client review and work quality. We are known as best commercial RO plant manufacturers, industrial RO plant manufacturer, sewage treatment plant manufacturer, Water Softener Plant Manufacturers and effluent treatment plant manufacturers. Apart from this 24x7 customer support is our USP. Call on +91-9650608473, or write us at enquiry@netsolwater.com for any support, inquiry or product-purchase related query.