HOW DOES WATER SOFTENER WORKS?
By using a simple ion exchange mechanism, the resin or mineral inside the mineral tank is precisely engineered to remove "hard" particles of lime and calcium. The dissolved particles in the entering water have a different or opposite electrical charge than the resin beads inside the softener tank. Because of the electrical charge difference, dissolved particles present in the water will attach to the resin beads when they come into touch with them, removing them from the water and making it "soft." The resin can only contain so many of these hardness particles, which is why softeners come in a variety of sizes and why regeneration or brining is required.
Discus the ion exchange process in water softening?
Aside from industry, ion-exchange softeners are widely used in small water systems and individual homes. Ion-exchange resins, exchanges one ion from the treated water for another ion in the resin (sodium is one component of softening salt, with chlorine being the other). Sodium is exchanged for calcium and magnesium by ion exchange resin. The presence of certain metal ions in water causes a wide range of issues. These ions disrupt the action of soaps. They also cause lime-scale buildup, which can clog plumbing and cause galvanic corrosion.
Traditional water-softening appliances for home use rely on an ion-exchange resin to exchange "hardness ions" – primarily Ca2+ and Mg2+ – for sodium ions. "Ion exchange devices reduce hardness by exchanging magnesium and calcium ions (Mg2+ and Ca2+) for sodium or potassium ions (Na+ and K+)."
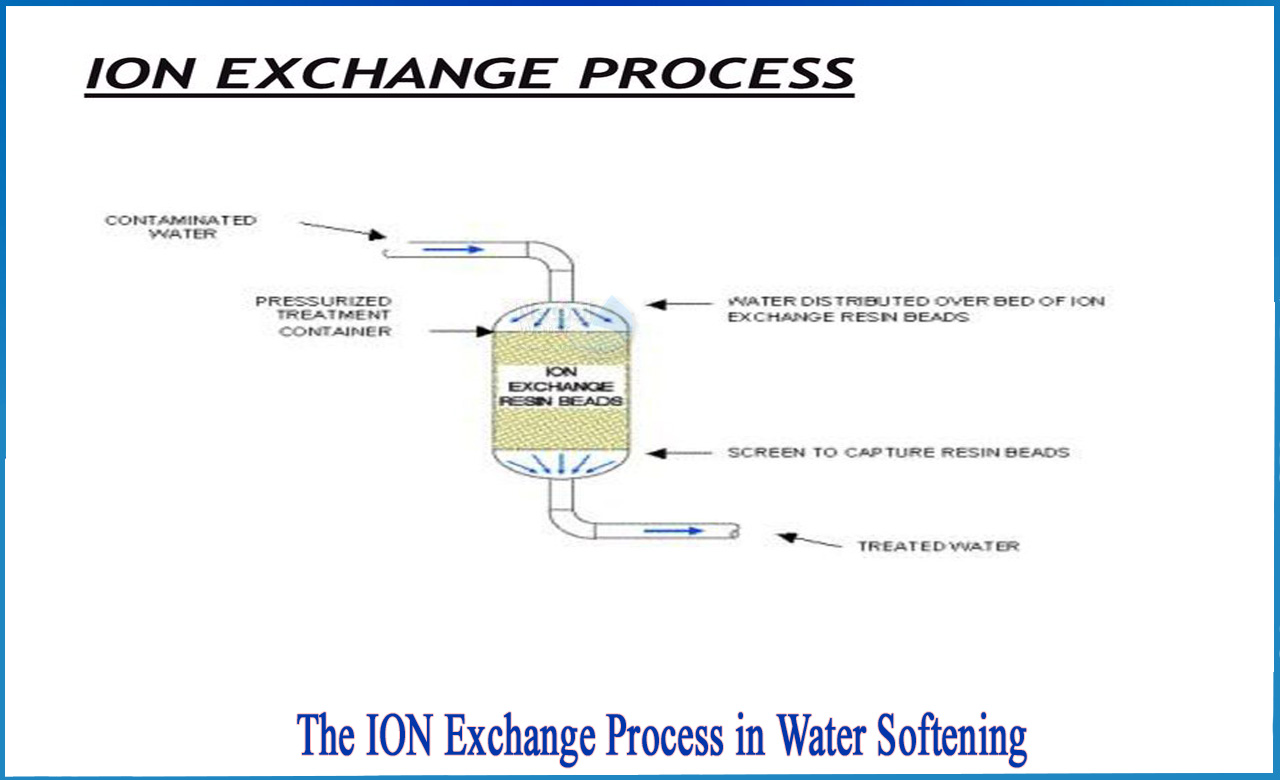
ION EXCHANGE UNITS
When all of the available Na+ ions have been replaced by calcium or magnesium ions, the resin must be re-charged by eluting the Ca2+ and Mg2+ ions with a sodium chloride solution. In the case of household units, the waste waters eluted from the ion exchange column containing the unwanted calcium and magnesium salts are typically discharged to the sewage system. These devices are similar to pressure filters. The interior is generally treated to protect the tank from salt corrosion.
The units are typically of the downflow type, and their size and volume are determined by the hardness of the water and the volume of treated water required between regeneration cycles. During regeneration, resin is supported by an underdrain system that removes treated water and evenly distributes brine. The minimum depth of resin should be 24 inches above the underdrain.
BACKWASH CYCLE
When hardness is reached, or the design output between regeneration (OBR) is reached, the softener must be regenerated. In down-flow units, the resin must be backwashed first to loosen it (it becomes compacted by the weight of the water) and to remove any other material that the resin has filtered out of the water. The operator must apply enough backwash water to expand the resin bed by approximately 50%. Distributors at the unit's top ensure uniform water distribution and wash-water collection. Underdrains ensure that the backwash water is distributed uniformly on the resin's bottom.
BENEFITS OF ION EXCHANGE SOFTENERS
Ion-exchange softening has some advantages over lime-soda ash softening. It is small and has a low initial investment. The chemicals used are less hazardous to the operator, and the operation is much simpler. It is entirely automatable.
Softeners can also be of the automatic duplex variety, which ensures a continuous supply of soft water in critical applications such as boiler feed. Many systems have discovered that ion-exchange is the most cost-effective method of producing high-quality water for their customers.
Netsol Water is Greater Noida-based leading water & wastewater treatment plant manufacturer. We are industry's most demanding company based on client review and work quality. We are known as best commercial RO plant manufacturers, industrial RO plant manufacturer, sewage treatment plant manufacturer, Water Softener Plant Manufacturers and effluent treatment plant manufacturers. Apart from this 24x7 customer support is our USP. Call on +91-9650608473, or write us at enquiry@netsolwater.com for any support, inquiry or product-purchase related query.