Overview
Many water treatment systems use a combination of coagulation, sedimentation, filtration, and disinfection to provide clean and safe drinking water to the public. The combination of coagulation, sedimentation and filtration is the most widely used water treatment technology in the world and has been in use since the early 20th century.
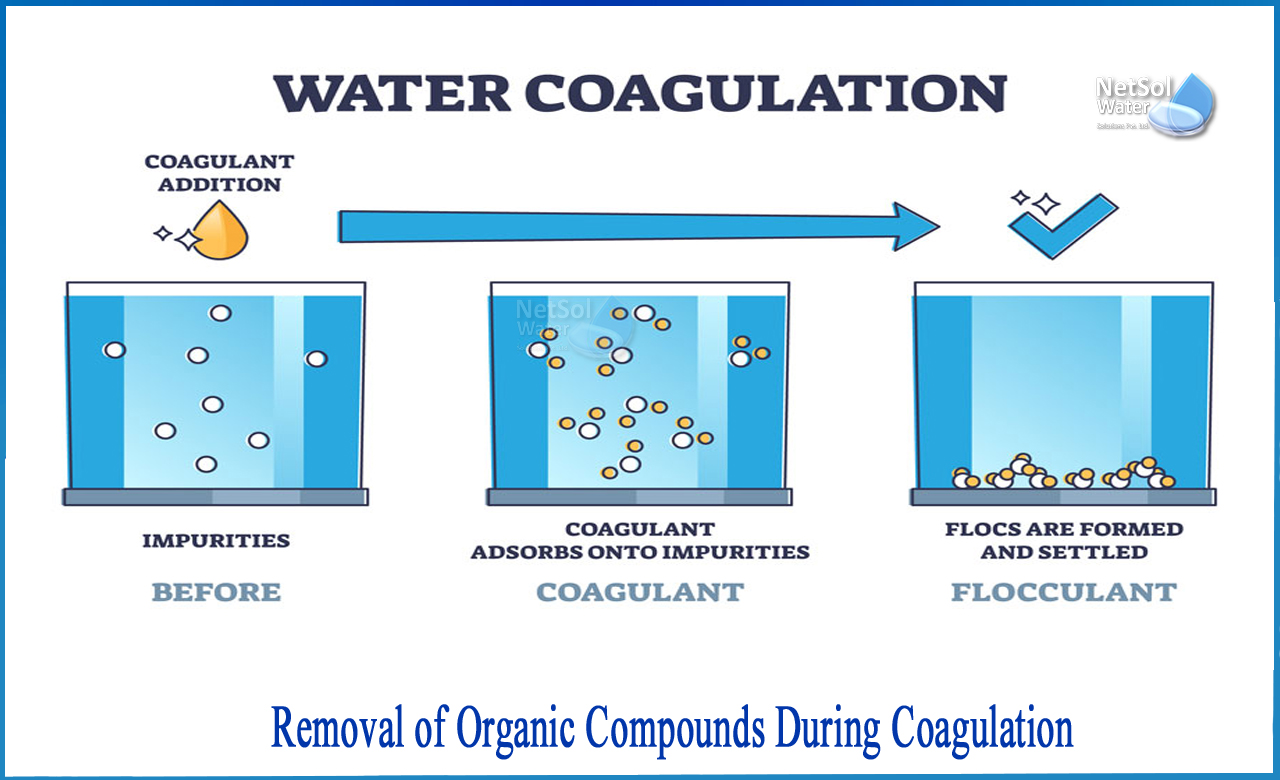
What is coagulation?
The solidification process involves the addition of aluminium salts such as iron or aluminium sulphate. These chemicals are called coagulants and are positively charged. The positive charge of the coagulant neutralizes the negative charge of the particles dissolved and suspended in water. During this reaction, the particles bind or coagulate to each other (this process is sometimes called agglutination). Large particles and flakes are heavy and quickly settle to the bottom of the water supply. This settling process is called settling.
In the water treatment plant, a coagulant is added to the water and the coagulant is quickly mixed to circulate the water. The solidified water can be directly filtered with a medium-sized filter (sand, gravel, etc.), microfiltration membrane, ultra filtration membrane, or supplied to the settling tank. In a settling or septic tank, heavy particles settle to the bottom and are removed, and water enters the filtration step of the treatment process.
What is removed during coagulation?
Coagulants can successfully remove large amounts of organic compounds, including some dissolved organic matter known as Natural Organic Matter (NOM) or dissolved carbon called Organic carbon (DOC). Coagulation can also remove suspended particles, including inorganic particles. Large amounts of DOC can give water an unpleasant taste, odourand browncolour. Coagulation can remove particles and some solutes, but water may contain pathogens.
Solidification removes many particles, such as dissolved organic carbon, making it difficult to disinfect water. Some of the dissolved material is removed by coagulation, so less chlorine needs to be added to disinfect the water. Because trihalomethanes (THMs) are dangerous by-products produced when chlorine reacts with NOM, municipal water treatment plants save costs by reducing chlorine usage and making water safer.
With accurate administration and correct application, the residue of added chemicals is usually not a problem!
Residues are by-products that remain in the water after the substance is added and the reaction takes place in the water. Each residue depends on the coagulant used. If iron (III) sulphate is used, iron and sulphate precipitates are added to the water. If you use aluminium sulphate, aluminium and sulphate is added to the water. Most urban water treatment plants use aluminium sulphate as a solidification chemical.
In general, water treatment plants set up the coagulation process so that coagulation chemicals are removed along with the flakes. However, treatment plants that use aluminium-based coagulants often have high levels of aluminium in the treated water, but not so often.
Conclusion
We need to coagulate waste using appropriate coagulating agents in order to satisfy effluent discharge demand from water treatment plants at economical prices.
Netsol Water is Greater Noida-based leading water & wastewater treatment plant manufacturer. We are industry's most demanding company based on client review and work quality. We are known as best commercial RO plant manufacturers, industrial RO plant manufacturer, sewage treatment plant manufacturer, Water Softener Plant Manufacturers and effluent treatment plant manufacturers. Apart from this 24x7 customer support is our USP. Call on +91-9650608473, or write us at enquiry@netsolwater.com for any support, inquiry or product-purchase related query.