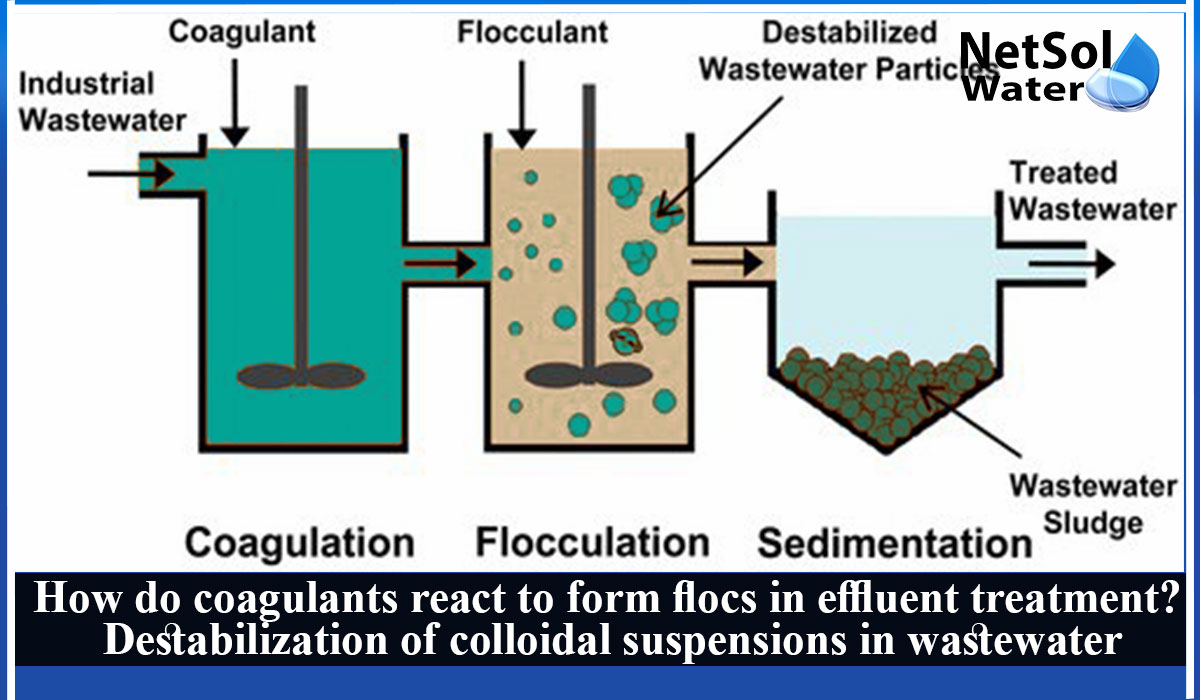
How does a coagulant work to create floc and Destabilization of colloidal suspensions in wastewater?
Numerous pollutants are found in wastewaters as colloidal dispersions, or as particles with sizes between about 1 and 10-3µm. Clays and biologically derived substances including natural colours, proteins, polysaccharides, and their synthetic or natural counterparts are examples of this type of suspension.
These types of suspensions almost have an innate stability or resistance to particle aggregation. Due to their extremely slow settling velocity, they cannot be clarified by sedimentation, and they also cannot be clarified using sand filtration techniques. Thus, the process of coagulation has an impact on their conversion to a flocculent state.
In this article, we will discuss about the chemistry of coagulants and flocculants, i.e., how coagulants react to form flocs in ETPs.
How do you destabilize colloids in wastewater?
Destabilization of colloidal suspensions in wastewater-
Colloidal particles have a very high specific surface area. As a result, surface characteristics dominate how they behave in suspension, with gravity effects on movement being comparatively minor. Clays and metal oxides, plant and animal residues, proteins, starch, detergents, etc., are allhydrophobic or hydrophilic colloidal particlespresent in wastewater.
Hydrophobic colloids in wastewater
These are thought to derive their stability from the presence of tiny particles, with like charges that resist one another and have no affinity for water. These charges could result from the particle surface's chemical composition, or from the preferential adsorption of a specific ion type. A colloid's charge, whether positive or negative, causes an "electrical double layer" to surround it, creating a potential gradient.

What is Zeta potential?
If all of the particles in a given colloidal dispersion have a similar charge, the electrostatic repelling forces stop the particles from coming together due to Brownian motion, and van der Waals attractive forces keep the suspension stable.
The potential at the rigid-solution border or the plane of shear can be measured, but not the potential at the solid-particle boundary. This latter is known as the zeta potential.
Relationship between zeta potential and coagulation
Zeta potential typically ranges from 20 to 200 mV. When the zeta potential is zero, the best coagulation may be anticipated to happen (the isoelectric condition of the suspension). Reduce the zeta potential to within 0.5 mV of the isoelectric point for efficient coagulation.
Hydrophilic colloids in wastewater
As their name suggests, hydrophilic colloids have a strong attraction for water. Although, charge is thought to play a role in their stability, their stability is mostly a result of bonded water layers that keep particles from coming into close contact with one another.
These organic compounds can exist as individual macromolecules, aggregates of macromolecules, or in suspension or real solution. They acquire their charge by the ionisation of functional groups, such as carboxyl (-COO-), hydroxyl (-OH-), sulphate (-SO3-), phosphate (-PO3H-), and amino (-NH3+) groups that are connected to them.
The degree of ionisation of the functional groups, which is affected by the medium's pH, determines the size of this charge. The solubility of hydrophilic colloids is also influenced by charge, and it is usually discovered that the isoelectric point, which is typically found in the pH range of 4.0 to 6.5, coincides with the minimal solubility.
Protective colloids and their relationship with coagulation
Hydrophobic colloidal particles like clays may absorb hydrophilic colloids, giving the latter hydrophilic characteristics. These colloidal suspensions are referred to as "protective colloids", and might be challenging to coagulate.
What are the main reasons that cause coagulation in wastewater treatment?
There are four main ways to cause coagulation, or the disintegration of colloidal stability:
(i) Boiling;
(ii) Freezing;
(iii) Reciprocal flocculation by adding a colloid of opposite charge; and
(iv) Adding electrolytes.
In the practise of water and wastewater engineering, only the latter is of significant importance.
Coagulation caused by the effect of electrolytes
Two possible actions of added electrolytes that tend to diminish zeta potential and therefore the stability, especially of hydrophobic colloids, can be proposed.
They diminish the thickness of the double layer by boosting the ionic strength of the dispersing medium, while a colloid's adsorption of opposing-charge ions lowers its own net charge.
The Schulze-Hardy rule, which states that "the precipitation of a colloid is effected by that ion of an added electrolyte, which has a charge opposite in sign to that of colloidal particles, and the effect of such ions increase markedly, with the number of charges it carries," was developed in response to the discovery that, the effectiveness of added ions of opposite charge, increases dramatically with their valency.

Influence of the added electrolytes on the colloidal stability
Chemical coagulation removes colloids through two more methods:
(a) Inter-particle bridging, and
(b) Enmeshment by the hydroxo-metal precipitate produced by the coagulant chemicals.
With increasing coagulant dose and thus rising volumetric concentration of enmeshing solid surface area, it is reasonable to predict that the relevance of the enmeshment process will increase.
Polyelectrolytes are especially linked to the inter-particle bridging mechanism.
Salts of aluminium and iron used in coagulation
The trivalent salts of aluminium and iron are the most significant coagulating agents, in the treatment of water and wastewater.
Its superiority as coagulants results from:
(a) Their efficiency in destabilising the primarily negatively charged colloids, present in natural waters and wastewaters,
(b) The generation of drinking water is particularly dependent on their low solubility levels, in the pH range of typical use, and
(c) They are readily available and reasonably inexpensive.

Coagulation with polyelectrolyte’s
Polymers with functional groups, such as carboxyl, hydroxyl, amino, and other groups, are known as polyelectrolyte’s. These charged sites give them characteristics similar to those of common low molecular weight electrolytes, such as solubility and electrical conductivity.
These could be categorised as hydrophilic colloids when in solution. They can be anionic, cationic, or ampholytic depending on the functional groups that are connected to them. The examples include:
Polyacrylate (_CH2_CH_CH2_)n anionic
Ι
COO-
Polyvinyl pyridinium (-CH2-CH-CH2-)ncationic
Polyaminoacids (_NH_CH_CO_NH_CH_CO_) ampholytic
Ι Ι
(CH2)4 (CH2)4
Ι Ι
(NH3)+ COO-
Mechanism of coagulation
According to theory, the mechanism of coagulation with polymers is mostly adsorptive and bridging, resulting in a loosely connected network of flocs. Although, it has been discovered that some polyelectrolyte’s are successful in coagulating colloidal dispersions, carrying a charge of the same sign as that held by the polyelectrolyte, neutralisation of charge may also play a role in the mechanism.
How do coagulants react and form flocs in effluent treatment?
Brownian motion causes initial floc particles, which are generated when a colloidal suspension is destabilised, to grow in size through contact with other particles. It is known as perikinetic flocculation.
When particles get bigger, Brownian motion effects have less of an impact, which slows down the pace of particle aggregation. Velocity gradients are produced within the dispersing fluid's body to quicken the rate of particle impact. This regulated application of velocity gradients to promote flocculation is often referred to as orthokinetic flocculation.
How does velocity gradient affect flocculation?
Colloidal particles must be brought close enough to each other to experience their van der Waals forces of attraction, in order to form aggregates.
Let's take a look at any suspended particle with a radius of influence of “Rj” as indicated by the van der Waals force. It will draw any other suspended particle “I” to it, so long as the centre of that particle is inside a sphere of influence with a radius of (Rj+Ri), around particle “j”.
Consider a streamtube with radius R = Rj+Ri and a velocity of “v” in the centre, and a constant velocity gradient in the z-direction. The formula for the volume of fluid moving past particle “j” in the stream tube is provided by:
Vol/time = 2?0Rz dv/dz bdz
Also,
G= dv/dz (velocity gradient)
It is necessary to avoid high velocity gradients since they have a tendency to shear floc particles. The recommended operating range for G is 30–60/.
Mixing methods and floc development
Commonly, concentrated solutions of coagulants are applied to a water or wastewater flow. A quick mixing system is necessary to guarantee the coagulant is distributed uniformly. A more moderate agitation is needed to create velocity gradients of a size sufficient for flocculation, after this first fast mixing.
The advantages of gravitational systems include their simplicity and ease of upkeep. Fast mixing can be produced at a hydraulic jump in an open channel or by injection into a turbulent pipe flow (velocity > 1 m/s).
Horizontal baffle basin for flocculation
To provide the mild amount of mixing necessary to induce flocculation, horizontal-flow basins with fixed baffles or upward-flow sludge blanket clarifiers may be utilised.
InahorizontalflowbaffledbasinofvolumeV(m3),withaflow-throughrateQ(m3s-1)andheadlossh(m),the powerperunitvolumeis
P/V=ρghQ/V
=rgh/t
Where,tisthedetentiontime(s).Thevelocitygradientis
G=(rgh/mt)0.5
Gt=(rght/m)0.5
The benefit of baffled basins is that energy dissipation is not evenly distributed; it is excessive at bends and insufficient on straights. They operate inflexibly as well.
Are you still confused about the process of coagulation and flocculation in effluent treatment plants? You do not need to worry!
Netsol Water is the leading manufacturer, supplier, and exporter of a quality selection of water treatment and wastewater treatment companies in delhi, India.
Reverse osmosis plants as well as water softeners, ETPs, DM plants, Ultra filtration, UV water purification systems, STPs, ZLD plants, AMC, O&M, and other goods and services are available from us. We also provide services to businesses including automotive, pulp & paper, beverages, pharmaceutical, textile, refineries, schools, hotels, hospitals, office buildings, among others.
Call us at +91 9650608473 or email at enquiry@netsolwater.comfor further information.