Explain the working of Activated Carbon Filters in detail
Activated carbon filtration is one of the procedures frequently used in water purification. Since, it can also remove odour-causing contaminants it is frequently used to make drinking water more pleasant, to consume. In this article, we will explain the working of activated carbon filters and its various uses.
Theory of Filtration and its evolution
For thousands of years, people have used filtration to remove rust, suspended particles, and other impurities from water. This is accomplished by putting the contaminated influent (input water) through a filtration medium. The filter media substance traps the impurities as the water flows through it.
Several different physical and chemical mechanisms are involved in removing impurities from the water, depending on the impurities and the media. Some of the equipment’s used for these processes have undergone significant changes over time. Our understanding of the basic physical and chemical processes involved in filtering has also improved. These developments have made it possible for experts in water purification, to maximise the removal of impurities from the water.
Why is activated carbon used for the process of water purification?
As we are aware that because of the huge surface area of the filter media, filtration systems not only remove particulate matter but can also be used to trigger chemical reactions, which remove a variety of contaminants.
Activated carbon or AC is a carbonaceous adsorbent with higher internal permeability, and an internal surface area. The interior surface area of commercial activated carbon grades ranges from 500 to 1500 m2/g.
There are three main categories of activated carbon that are used for water purification:
· Particle sizes of activated carbon powder which range from 1 to 150 m.
· 0.5 to 4 mm-sized granular activated carbon particles.
· Activated carbon extruded with 0.8–4 mm-sized particles.
How to choose the correct activated carbon for your water treatment application?
The right activated carbon has several distinctive qualities, including an internal surface area, specific (surface) chemical properties, and excellent accessibility of internal pores. The best fit relies on the molecules to be trapped, the phase (gas, liquid), and the treatment conditions. The pore size distribution is also crucial for practical application. Therefore, combining the appropriate raw material and activation circumstances, results in the desired pore structure of an activated carbon product.
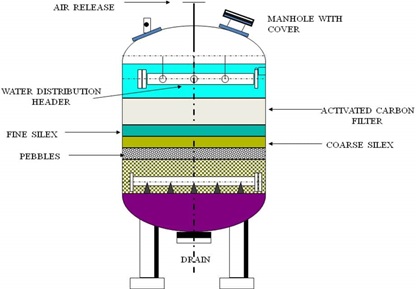
Working principle of activated carbon
One of the most commonly used but least understood words in filtration discussions is "adsorption." Adsorption is the process by which a contaminant is transferred from a liquid to the surface of a solid. When adsorption takes place, a water-born, floating particle sticks to a solid surface.
Atoms, electrons, or molecules from a gas, liquid, or solid adhere to a surface through a process known as adsorption. When it comes to water filtering, the liquid's suspended solid particles will stick to the media's solid surface.
How does activated carbon work?
In most cases, activated carbon works by adsorbing impurities from liquids, gases, or vapours. It is crucial to match the pore size of the activated carbon media with the molecules and particles you are trying to adsorb, because this happens in pores that are just a little bit larger than the molecules that are being adsorbed.
Working of activated carbon in water purification systems
Anthracite or bituminous coal and coconut shells are biological sources of activated carbon. When an organic source is burned without air, carbon is produced. Only about 30% of the organic mass remains after this procedure, and this eliminate heavy organic molecules.
The organic material must then be "activated" before it can be used to treat water. The carbon's enormous numbers of pores are opened up during activation, which also further chases away undesirable molecules. The open pores are what enable the carbon to capture contaminants.
Methods of activated carbon activation
Two primary trigger?ing techniques exist:
1: Steam activation method
It is done using steam at temperature between 800 and 1000 degrees Celsius. The carbonised material gasifies instantly at these temps due to the Water-Gas reaction. The vapours are then burned off without burning the carbon with the addition of air.
This procedure yields activated carbon that has been evaluated, screened, and dusted. The small pore structure of carbon that has been activated by steam, makes it perfect for adsorbing both liquid phase and vapour phase substances.
2: Chemical activation method
With chemical activation, a strong dehydrating agent is first added to the carbon, usually in the shape of phosphoric acid (P2O5) or zinc chloride paste (ZnCl2). The carbon is activated by heating the mixture to a range of 500°C and 800°C.
The wide pore structure of activated carbon that results from chemical activation, makes it better suited, for adsorbing large molecules.
Uses of activated carbon in water purification
There are essentially two uses for activated carbon water purification, and each one operates in a completely unique manner.
1. Chlorine removal: It can be accomplished using activated carbon with less deterioration or harm to the carbon. Rapid de-chlorination and high flow rates are common.
However, a large quantity of surface area is needed for this process, and eventually water organics will fill up and block the carbon's pores.
2. Organic matter: These are removed by the "adsorption" process, which traps particles and compounds inside an activated carbon filter as water flows through it.
Factors which affect adsorption process in the purification of water
The following crucial variables affect the adsorption process:
1) The surface area and pore size distribution of the activated carbon;
2) The amount of hydrogen and oxygen in the carbon source;
3) The chemical composition and concentration of the contaminant;
4) The pH and temperature of the water; and
5) The amount of time the water is exposed to the activated carbon filter (called empty bed contact time or EBCT)
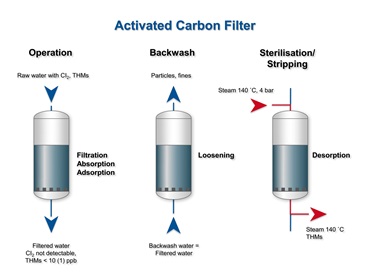
Applications of activated carbon filter (ACF)
Activated carbon filters have a wide range of uses. Some of them are mentioned below:
· Free elimination of chlorine
· Removing organic matter
· Odour elimination
· Removing bromate (After Ozonation of SWRO Permeate)
· Color removal from Sugar dissolving (White Sugar Manufacturing)
· Molasses coloration removal
Features of activated carbon filter
· The filter is made up of different types of carbon granules that are held in place by gravel, pebbles, and sand.
· Depending on the sort of treatment needed, different grades of carbon are used.
· The best iodine value for efficient contamination elimination can be found in activated carbon.
· Long-lasting activated carbon for dechlorination, organic contaminant elimination, and colour removal with a 900 Iodine value.
· Offers consistently high-quality purified water.
· Pressure containers made of non-corrosive FRP and have UPVC front piping.
· Designed to operate between 3.5 and 5.0 kg/cm2 of pressure with a minimum input TSS of 5 ppm.
How can we help you?
To satisfy particular process needs, Netsol Water Solutions’ provides a broad selection of activated carbon filters (ACF) for various applications. Our ACFs are used in effluent treatment plants, sewage treatment plants, and water treatment plants, as second stage filtration following MGF/PSF.
Pulp & paper, automotive, food, beverage, textiles, pharmaceutical, refineries, and commercial buildings like hotels, schools, office buildings, hospitals, etc. are a few of the industries we serve. For further information, contact us by phone at +91 9650608473 or by email at enquiry@netsolwater.com



