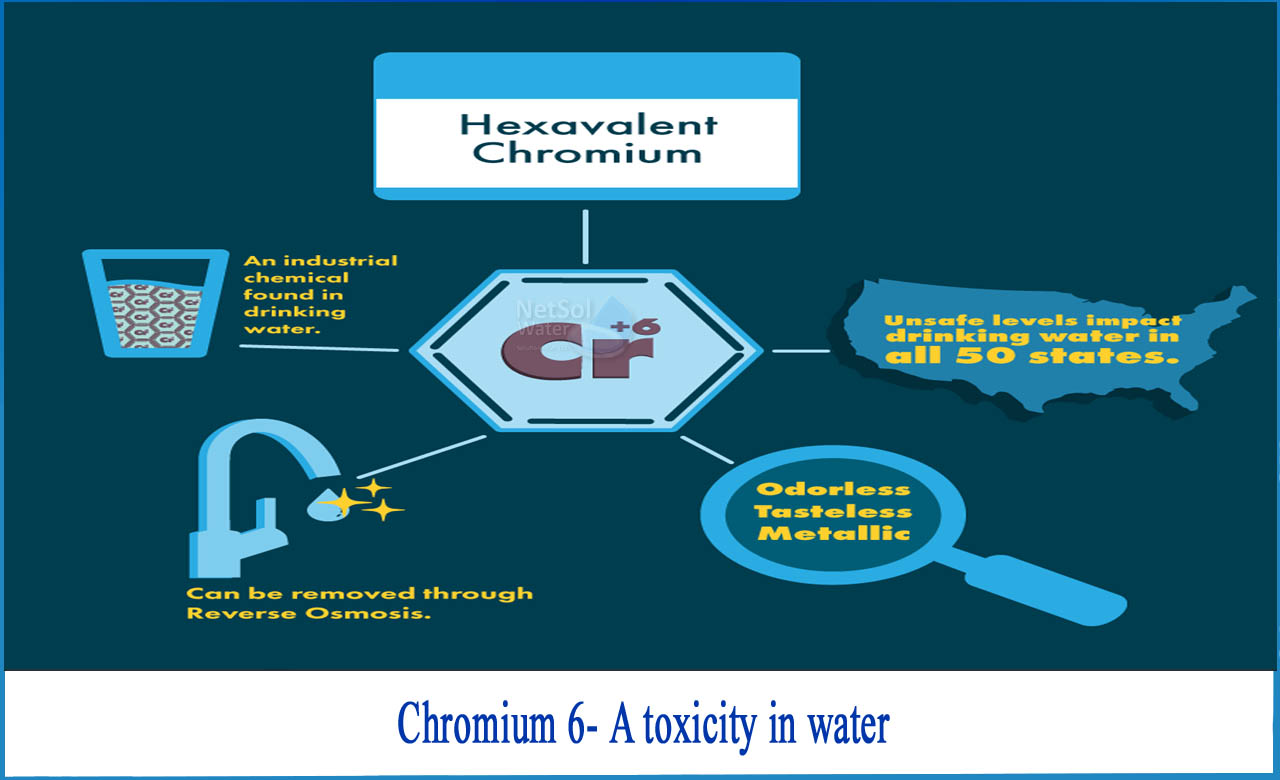
What is Chromium-6 and why is it a cause of concern?
Chromium occurs mainly in two forms in the environment. It is trivalent chromium (chromium 3), which is an essential nutrient for humans, and hexavalent chromium (chromium 6), which is toxic. Despite this difference, chromium in drinking water is regulated as "total chromium" with a content of 50 ppb.
These standard averages the toxicity of hexavalent chromium in water and trivalent chromium, which is less dangerous. The combined approach is clearly inadequate to protect public health, as state scientists have found that hexavalent chromium levels in drinking water that do not have a significant health impact are 0.2 ppb.
Unfortunately, in mostplaces in India, people do not bother what is dissolved in their drinking water. Water is polluted and lives and health are at risk, including school children who do not have access to bottled water.
As per EPA, Chromium is listed on the top 14 harmful chemicals and in India the permissible levels of chromium in as per IS 10500 is 0.05 mg/l.
What are the sources of hexavalent chromium?
In addition to natural sources, hexavalent chromium enters drinking water sources through colour and colour pigments, wood preservatives, chrome-plated waste emissions, and leaching of hazardous waste from landfills. Not surprisingly, communities near chrome waste landfills or chrome manufacturing and treatment facilities are at particular risk of exposure. Perhaps the most affected are the workers exposed in the workplace.
Who is exposed to hexavalent chromium?
Many workers in various occupations are potentially exposed to hexavalent chromium. Occupational exposure mainly occurs in the following areas:
Chromium metal is added to alloy steel to improve hardenability and corrosion resistance. The main cause of workers' exposure to Cr (VI) occurs during "hot work" such as in the workplace. Welding to stainless steel and other alloy steels including chrome metals. Cr (VI) compounds can be used as pigments in dyes, paints, inks, and plastics. It can also be used as an anticorrosive agent added to paints, primers, and other surface coatings. Chromic acid, a Cr (VI) compound, is used to electroplating chromium on metal parts to provide a decorative or protective coating
What are the health effects of hexavalent chromium?
Hexavalent Chromium [Cr (VI)] is one of the valence levels (+6) of elemental chromium. It is usually made by an industrial process. Cr (VI) is known to cause cancer. In addition, it acts on the respiratory tract, kidneys, liver, skin, and eyes.
Hexavalent chromium is carcinogenic and has reproductive toxicity in men and females.
Exposure to hexavalent chromium occurs by inhalation, ingestion, and skin contact. Most of the known health effects can be traced back to inhalation, but there are now strong data linking hexavalent chromium intake, for example through drinking water, to serious health effects. In addition to cancer and reproductive disorders, short-term and long-term exposures include eye and respiratory irritation, asthma attacks, nasal ulcers, skin burns, anaemia, acute gastroenteritis, dizziness, gastrointestinal bleeding, convulsions, ulcers, and liver. It may cause damage to the bodyas well as kidney.