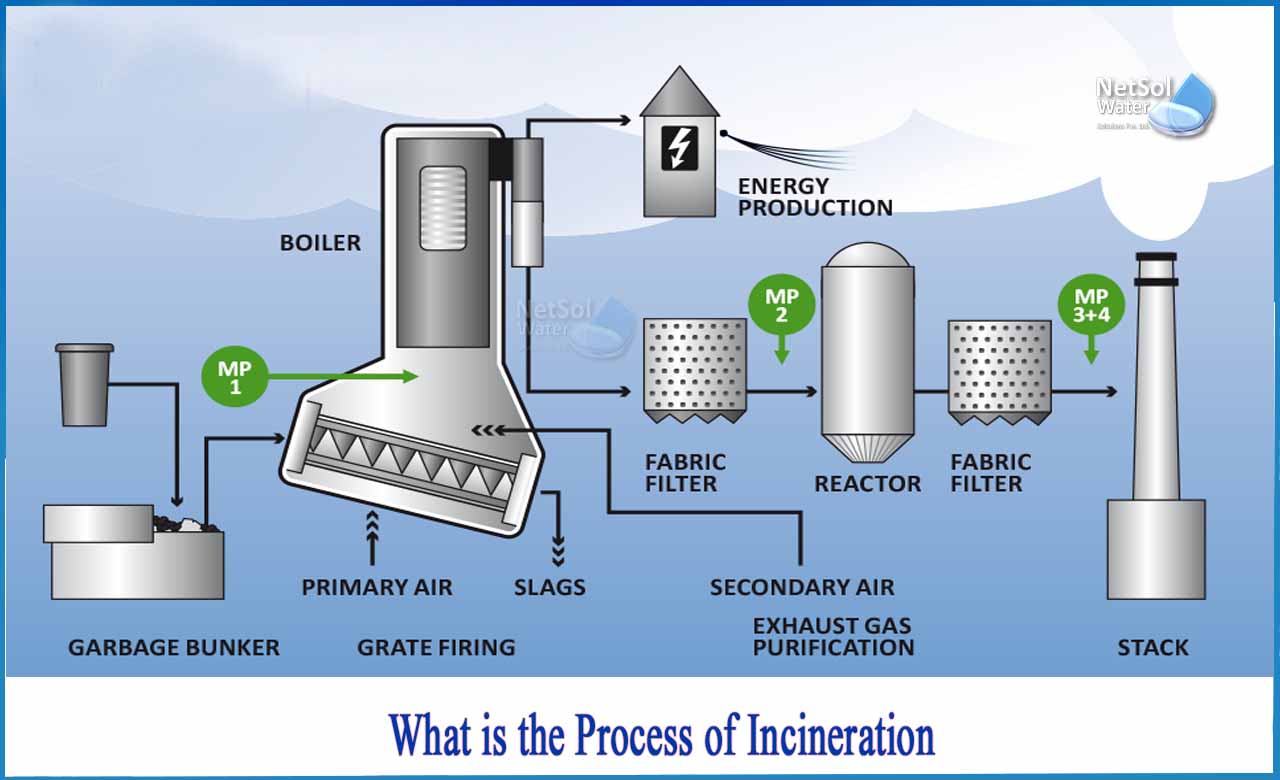
What is the process of incineration?
Incineration is a waste treatment procedure that involves the burning of organic compounds contained in waste products. The original waste's solid bulk is decreased by 80 to 85 percent, while the volume is lowered by 95 to 96 percent. While incineration does not completely replace landfilling, it does significantly reduce the amount of garbage that must be disposed of. Not only that, but when utilized to treat more specialized forms of garbage, such as clinical or hazardous waste, high temperatures may remove potentially dangerous poisons and germs.
Pollutants are produced when garbage is burned. Smaller amounts of CO, NOx, HCl, HF, HBr, PCDD/F, SO2, VOCs, PCBs, or heavy metal compounds are generated or persist after combustion, depending on the nature of the material being burnt.Most nations control these emissions and have environmental legislation in place.
Particular attention should be paid to HCl, which must be appropriately controlled not just owing to possible environmental harm, but also due to the risk it poses to plant equipment. Wastes frequently contain chlorinated organic compounds or chlorides, with PVC accounting for over half of these chlorides in municipal garbage. The organic component of these compounds is eliminated during the incineration process, and any chlorine present is transformed into HCl.
This problem generally affects the following plant species:
• Waste incinerators, such as those used to dispose of hazardous, municipal, and sewage sludge waste.
• Waste-incineration plants, such as power plants, cement factories, or biomass facilities.
Techniques for Reducing Acid Gases
Alkaline reagents are typically used to remove HCl, SO2, and HF from flue gas. The following procedures are used:
Dry processes: The flow of flue-gas is treated using a dry sorption agent (such as sodium bicarbonate or lime). The reaction's byproduct is also dry.
Semi-wet processes: These processes are also known as semi-dry processes. The sorption agent in this case is a suspension (such as a slurry) or an aqueous solution (such as lime milk). The water solution evaporates, leaving dry reaction products behind. Any leftover can be recirculated to increase reagent use. Fly-dry procedures are another version of this method. An injection of water here provides rapid gas cooling, and reagent at the filter input.
Wet processes: The flue-gas flow is supplied into hydrogen peroxide, water, and/or a cleaning solution containing a portion of the reagent in this process (for example sodium hydroxide solution). The reaction product is aqueous in nature.
What may I dispose of in an incinerator?
Even if the ash is disposed away, burning anything does not completely remove it. A portion of the original waste is transformed to carbon dioxide and discharged into the environment. However, this pollution may be preferable to the alternative. If the same amount of garbage were disposed of in landfills, the resulting emissions of methane, a more potent greenhouse gas, would be considerably worse for the environment.
Researchers must contend with the following constraints:
>get solid predictions about the effect of scaling various setups from laboratory to semi-industrial or industrial dimensions;
>get trustworthy experimental data on huge sizes using statistically successful treatments;
>encourage the use of residual mixtures in the manufacture of high-quality goods via the application of experimental and numerical approaches connected by validation and uncertainty quantification tools;
>create novel catalytic materials capable of promoting efficient reactions while producing undesirable by-products;
>optimize process setups to reduce heat losses and enhance energy recovery efficiency;
>more accurately estimate pollutant emissions by a better knowledge of the underlying mechanisms that create NO2 and other dangerous sorts of emissions in these types of operations.
Netsol Water is Greater Noida-based leading water & wastewater treatment plant manufacturer. We are industry's most demanding company based on client review and work quality. We are known as best commercial RO plant manufacturers, industrial RO plant manufacturer, sewage treatment plant manufacturer, Water Softener Plant Manufacturers and effluent treatment plant manufacturers. Apart from this 24x7 customer support is our USP. Call on +91-9650608473, or write us at enquiry@netsolwater.com for any support, inquiry or product-purchase related query.