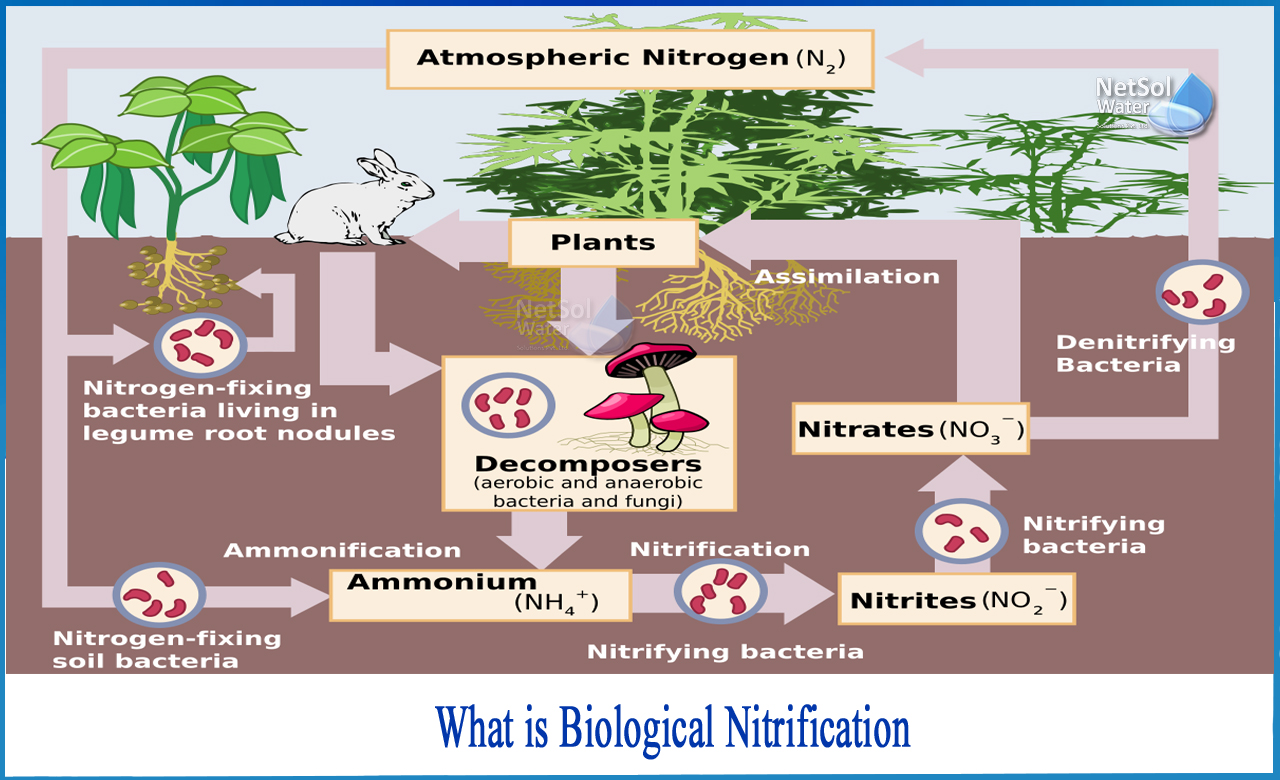
What is biological nitrification?
Nitrification is the biological oxidation of ammonia to nitrite, followed by nitrite oxidation to nitrate by separate organisms or direct ammonia oxidation to nitrate by comammox bacteria. The conversion of ammonia to nitrite is typically the rate limiting step in the nitrification process. Nitrification is a critical step in the nitrogen cycle of soil. Nitrification is an aerobic process that is carried out by small communities of autotrophic bacteria and Achaea.
The process followed
Nitrification is a microbial process that converts reduced nitrogen compounds (primarily ammonia) to nitrite and nitrate. Ammonia can be found in drinking water due to naturally occurring processes or by adding ammonia during secondary disinfection to form chloramines. The nitrification process is primarily carried out by two types of autotrophic nitrifying bacteria, which can construct organic molecules using energy derived from inorganic sources, in this case ammonia or nitrite.
Nitrogen removal occurs in conventional activated sludge systems to varying degrees (20-30%). Nitrogen removal rates of 70 to 90 percent can be achieved in plants based on activated sludge and other suspended growth systems using the nitrification-denitrification method. Prior nitrification of all ammonia and organic nitrogen in the incoming waste is required for biological denitrification.
There are two types of chemoautotrophic bacteria that can be linked to the nitrification process.
1: The energy for one group (Nitrosomonas) comes from the oxidation of ammonium to nitrite, while the energy for the other group (Nitrobacter) comes from the oxidation of nitrite to nitrate. Both groups, collectively known as Nitrif?iers, obtain the carbon they require from inorganic carbon forms.
The process of converting ammonia to nitrate consists of two steps:
· Stoichiometrically, 1 kg of nitrogen requires 4.6 kg of oxygen to be nitrified. Experiment evidence has shown that nitrite accumulation is insignificant under steady-state conditions. This suggests that the genus Nitrosomonas' oxidation of ammonium to nitrite is the rate-limiting step in the conversion of ammonium to nitrate.
· qc = 1/m,
Where “m” is the Nitrosomonas growth rate at the worst operating temperature. If nitrification is desired, the sludge age (or mean cell residence time), and
“qc” in a treatment plant must be sufficiently high.
Biological Oxidation and Nitrification Systems, Both Combined
Flow sheets for combined and separate systems of biological oxidation and nitrification are shown in the figure below:
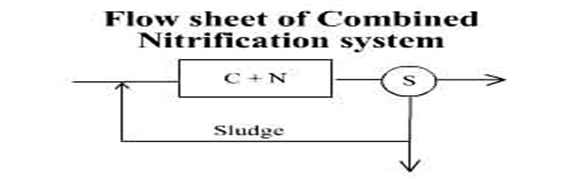
The combined system is the preferred method of operation because it is less sensitive to load variations (due to larger aeration tanks) and produces a smaller volume of surplus sludge (due to higher qc values used) and better sludge settleability.
Conclusion
It is critical to ensure that the aeration tank's oxygenation capacity is sufficient to meet the oxygen uptake due to carbonaceous demand and nitrification. Sludge recycling must be fast enough to prevent denitrification (and rising sludge) caused by anoxic conditions in the settling tank.
The first tank in a separate system can be smaller in size because a higher F/M ratio can be used, but this makes the system more sensitive to load variations and produces more sludge for disposal. To keep the two sludge’s separate, an additional settling tank is required between the two aeration tanks. The main advantage of this system is its higher nitrification efficiency and better performance when toxic substances are suspected to be present.
Netsol Water is Greater Noida-based leading water & wastewater treatment plant manufacturer. We are industry's most demanding company based on client review and work quality. We are known as best commercial RO plant manufacturers, industrial RO plant manufacturer, sewage treatment plant manufacturer, Water Softener Plant Manufacturers and effluent treatment plant manufacturers. Apart from this 24x7 customer support is our USP. Call on +91-9650608473, or write us at enquiry@netsolwater.com for any support, inquiry or product-purchase related query.