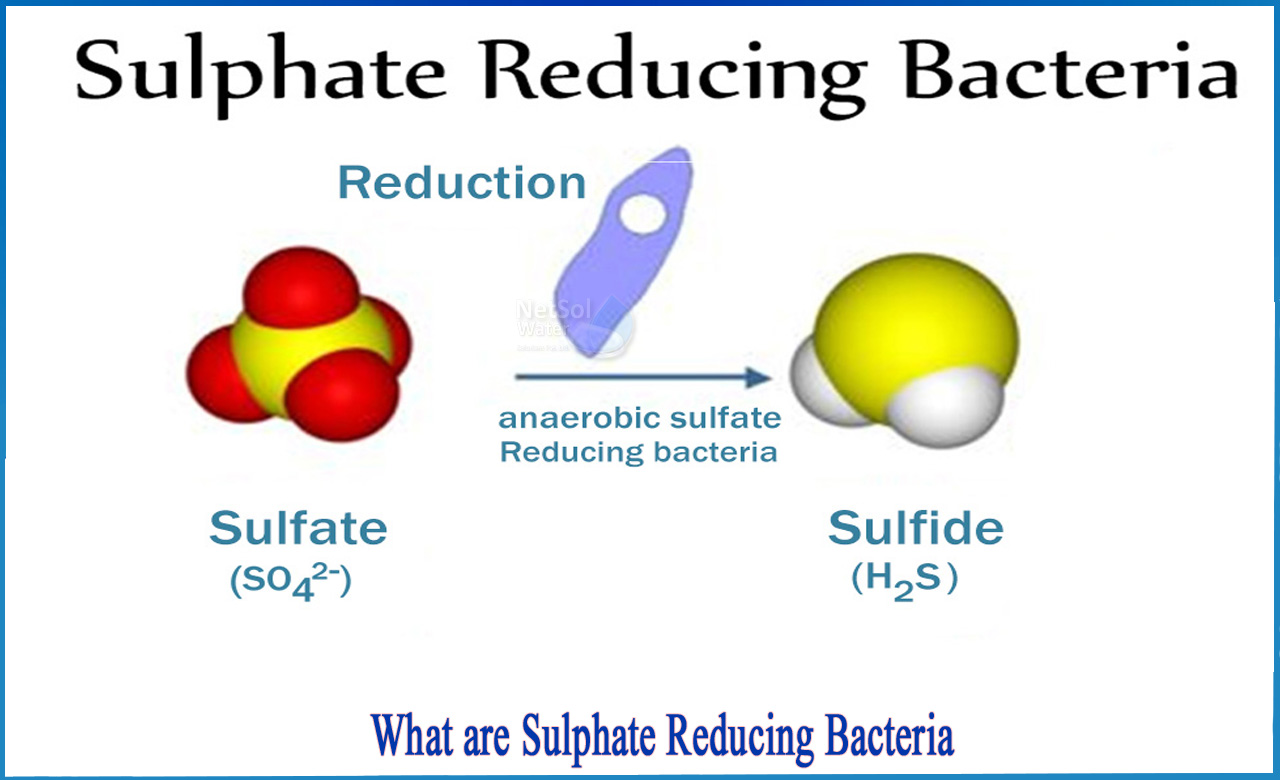
What are sulphate reducing bacteria?
Sulphur is a smooth, bright yellow element that is also known as brimstone because it burns, producing sulphur dioxide (SO2) and emitting a distinctive odour. It is linked to volcanoes and minefires.
In India, some near-surface coal mine fires emit sulphur vapour, which condenses on the underside of loose rock. The bright yellow sulphur deposits can be seen if you flip the rock over. Sulphur does not normally remain in its elemental state. Because the element, sulphur, is essentially insoluble in water, it does not pose a problem in drinking water.
In compounds, sulphur has many oxidation states, but the most popular and interesting to us are –2, –1, and +6. Sulphur will be lowered to an oxidation state of –2 or –1 in the absence of oxygen and will form sulphide compounds. The sulphur in pyrite has an oxidation state of –1, whereas the sulphur in hydrogen sulphide (H2S) has an oxidation state of –2. If there is plenty of oxygen, sulphur will be oxidised to an oxidation state of +6 and form sulphate (SO4) compounds like gypsum (CaSO4•2H2 O) and sulphuric acid (H2 SO4).
In low oxygen environments, sulphides are stable, whereas sulphates are stable in high oxygen environments. Things get interesting when sulphides are exposed to a high oxygen environment or when sulphates move into a low oxygen environment because they can end up in water by modifying to a more stable form in the new environment.
With one notable exception, sulphides are extremely insoluble in water. If they are exposed to air, they will weather, oxidise, and release sulphate anions into the water. Pyrite, a sulphide mineral, is frequently found in association with coal. Coal mining fractures the pyrite, and much of it ends up in rock waste piles (known locally as culm banks) where it is exposed to air and water. In this oxygen-rich environment, the pyrite degrades, releasing large amounts of dissolved iron and sulphuric acid into the mine water; sulphuric acid (H2 SO4) is the acid in acid mine drainage. In water, the acid quickly and completely dissociates to form H+ (the acid part) and the sulphate anion (SO4). The acid can be neutralised, but the sulphate is still present in solution.
Sulphate reducing bacteria
Certain bacteria can benefit from sulphur oxidation or reduction because such chemical changes provide energy. When sulphate-rich water enters a low-oxygen environment, sulphur-reducing bacteria thrive. Such bacteria mediate the transformation of sulphate into hydrogen sulphide, which, as a gas, can dissolve in water; sulphides, on the other hand, are very insoluble in water. Sulphur-oxidizing bacteria, on the other hand, derive energy from the oxidation of sulphides to sulphates in oxygen-rich environments.
Sulphur can then cause a variety of problems in drinking water, including sulphate and hydrogen sulphide gas, as well as various sulphur bacteria that aid in their production and insoluble sulphide particles suspended in the water.
Are sulphur reducing-reducing bacteria problematic?
Sulphur-reducing bacteria that use sulphur as a source of energy are the primary producers of large amounts of hydrogen sulphide. These bacteria use a chemical reaction to convert natural sulphates in water to hydrogen sulphide. Deep wells, plumbing systems, water softeners, and water heaters are examples of oxygen-deficient environments where sulphur-reducing bacteria thrive.
These bacteria are most commonly found on the hot water side of a water distribution system. Sulphur-oxidizing bacteria have similar effects to Iron Bacteria. They transform sulphide to sulphate, resulting in a dark slime that can clog pipes and/or stain clothing. A sulphur-oxidizing bacteria problem may be indicated by blackening of water or dark slime coating the inside of toilet tanks. Sulphur-oxidizing bacteria are rarer than sulphur-reducing bacteria.
Using a Water Softener and RO plant can help solve your problems from such bacteria. For more information, contact Netsol Water.
Netsol Water is Greater Noida-based leading water & wastewater treatment plant manufacturer. We are industry's most demanding company based on client review and work quality. We are known as best commercial RO plant manufacturers, industrial RO plant manufacturer, sewage treatment plant manufacturer, Water Softener Plant Manufacturers and effluent treatment plant manufacturers. Apart from this 24x7 customer support is our USP. Call on +91-9650608473, or write us at enquiry@netsolwater.com for any support, inquiry or product-purchase related query.