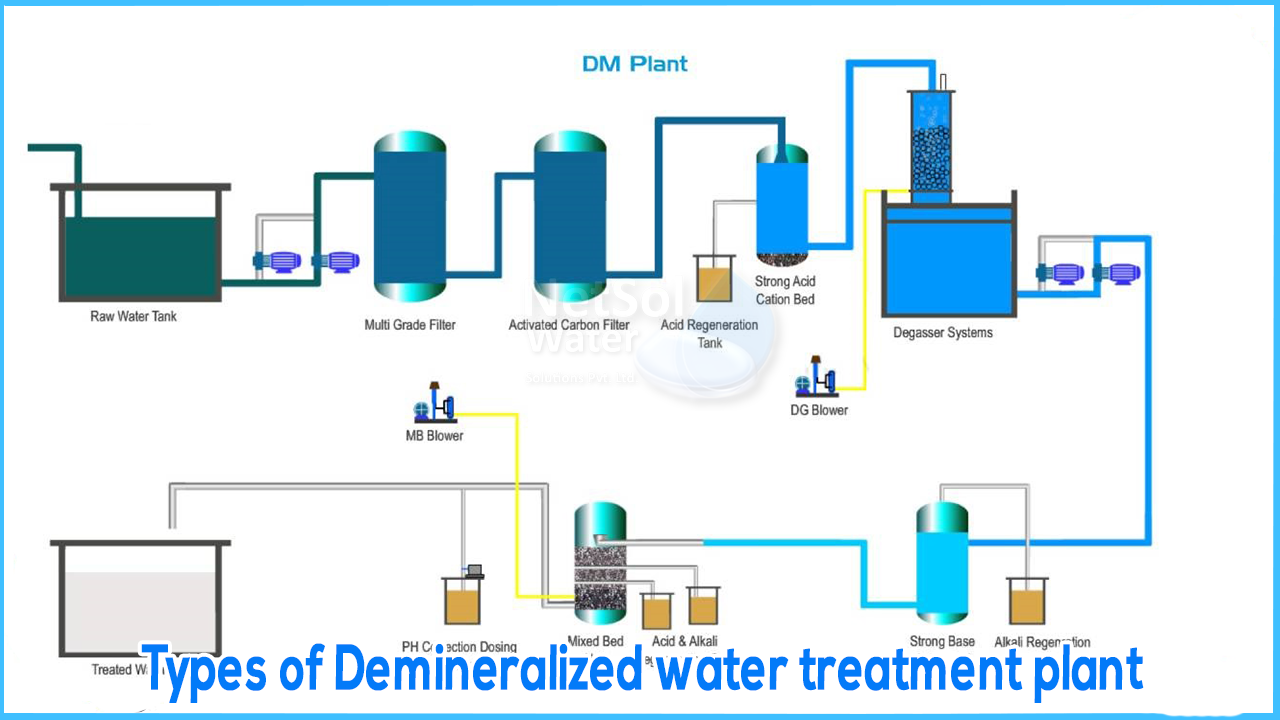
Types of Demineralized water treatment plant
Water that hasits mineral ions removed is known as Demineralized Water or Deionized Water. Mineral ions such as sodium, calcium, iron, copper, and others, as well as anions such as chloride, sulphate, and nitrate, are common ions found in water. Deionization is a physical process that involves the use of specially made ion exchange resins to offer an ion exchange site for the replacement of mineral salts in water with water, resulting in the formation of H+ and OH- ions.
Deionization provides a high purity water that is generally similar to distilled water, and the process is rapid and without scale formation because the bulk of water contaminants are dissolved salts. De-mineralization technology is a tried-and-true method of water treatment. A DM water system works on the principles of ion exchange, degasification, and polishing to provide mineral-free water. In the fields of steam, power, process, and cooling, the demineralized water system is widely used.
Ion Exchange Resins
Ion Exchange Resins are a type of resin that is used to exchange ions. Cation-exchange and anion-exchange resins are the two most common forms of resin. In exchange for impurity cations present in the water, cation exchange resins will release hydrogen (H+) ions or other positively charged ions. In exchange for impurity anions present in the water, anion exchange resins will release hydroxyl (OH-) ions or other negatively charged ions.
Ion-exchange technology can be used in water treatment and purification in three ways:
- 1. First, cation-exchange resins alone can be used to soften water through base exchange
- 2. Second, anion-exchange resins alone can be used for organic scavenging or nitrate removal
- 3. Third, a combination of cation-exchange and anion-exchange resins can be used to remove virtually all ionic impurities present in the feed water, a process known as “The purifying process of water deionizers” producing extremely high-quality water.
Let’s give a read to various types
Deionization
High-purity water that is virtually free of ionic pollutants is required for many laboratory and industrial applications. Deionization can be used to create water of this quality. The following are the two most prevalent types of deionization:
• Deionization (Two bed)
• Deionization (Mixed bed)
- 1. Two-bed deionization
The two-bed deionizer consists of two containers, one of which contains a hydrogen (H+) cation-exchange resin and the other of which contains an anion resin in the hydroxyl (OH-) form. All of the cations are exchanged for hydrogen ions as water travels through the cation column. To keep the water electrically balanced, one hydrogen ion is exchanged for every monovalent cation, such as Na+, and two hydrogen ions are exchanged for every divalent cation, such as Ca2+ or Mg2+. When it comes to anion-exchange, the same idea applies.
After that, the de-cationized water passes via the anion column. This time, all of the negatively charged ions are replaced with hydroxide ions, which join with the H2O
- 2. Mixed-bed deionization
Cation-exchange and anion-exchange resins are intimately combined and held in a single pressure vessel in mixed-bed deionizers. A mixed-bed deionizer is equal to a long sequence of two-bed plants due to the full mixing of cation-exchangers and anion-exchangers in a single column. As a result, the water quality produced by a mixed-bed deionizer is significantly superior to that of a two-bed machine.
Mixed-bed plants are more sensitive to pollutants in the water supply and have a more difficult regeneration process, although being more efficient at purifying the incoming feed water.After the water has been treated by either a two-bed deionizer or a reverse osmosis unit, mixed-bed deionizers are typically used to 'polish' it to greater degrees of purity.
Electro-deionization or EDI
Typically used in conjunction with reverse osmosis (RO) and other purification equipment, electro-deionization systems remove ions from aqueous streams.
Our high-quality deionization modules produce ultrapure water up to 18.2MW/cm on a continuous basis.