INTRODUCTION
Silica, often known as silicon dioxide or SiO2, is a crystalline substance found in nearly all rocks, minerals, and sand. Silicon and oxygen combine with another metal or mineral to make silica. Its concentrations range from one part per million to over one hundred parts per million. In most water supplies, silica is found in two forms: reactive silica and colloidal silica. On glassware and other fixtures, silica produces etching, scratching, and staining.
The removal of silica is frequently done to protect equipment from wear and damage. This is because silica can build up on surfaces and form scales, which is a hard mineral deposit. Scale deposits limit heating and cooling performance and make it difficult to run systems within a specified pressure range. The efficiency, performance, and safety of water-using equipment such as boilers, turbines, pipes, and even filtration systems can all be harmed by silica scaling.
Hardness is frequently confused with silica. Because of staining, it may appear that there is still hardness in the water after a water softener is installed. You can tell if it's hardness or silica by doing a simple test. Simply put vinegar to the spots, and if the vinegar removes them, the problem is hardness. It's most likely silica if the vinegar doesn't get rid of it.
The removal of pollutants from water, such as silica, can be a difficult task. Cooling towers, boiler blow feed water or boiler blow down water, carwashes, semi-conductor manufacturers, and steam injection oil recovery wastewater are some examples of sectors that require to remove silica from water before disposal or reuse. In fact, eliminating silica from process water is a challenge for any sector that processes wastewater for discharge or reuse.
REMOVAL OF SILICA
The most common source of silica in water is naturally occurring silica. Before treatment, reuse, or disposal/discharge, silica must be removed in whatever form it exists. Lime softening, ion exchange, and reverse osmosis are the most common ways for extracting silica from a waste stream.
1. LIME SOFTENING
Lime softening is a typical method for eliminating silica from water that is used to build up cooling towers, boilers, or boiler blow down water. Lime softening is a process that uses the addition of lime (calcium hydroxide) to precipitate hardness ions (calcium carbonate and magnesium carbonate). In flakes of magnesium and calcium hydroxides, silica particles are absorbed. The separated silica can subsequently be disposed of by sending these flakes (floc) through a clarifier or filter. The percentages of chemicals that must be added must change as the proportion of silica in the water changes.
2.ION EXCHANGE
Ion exchange is an excellent method for removing silica, however, the resin exchangers must be refilled and replaced on a regular basis, which adds major ongoing expenditures to the operation. Furthermore, if heavy metals are present in the exchange resins with the silica, these concentrated ion metals usually fail the TCLP leachability test and must be disposed of in a hazardous categorized landfill, incurring additional costs.
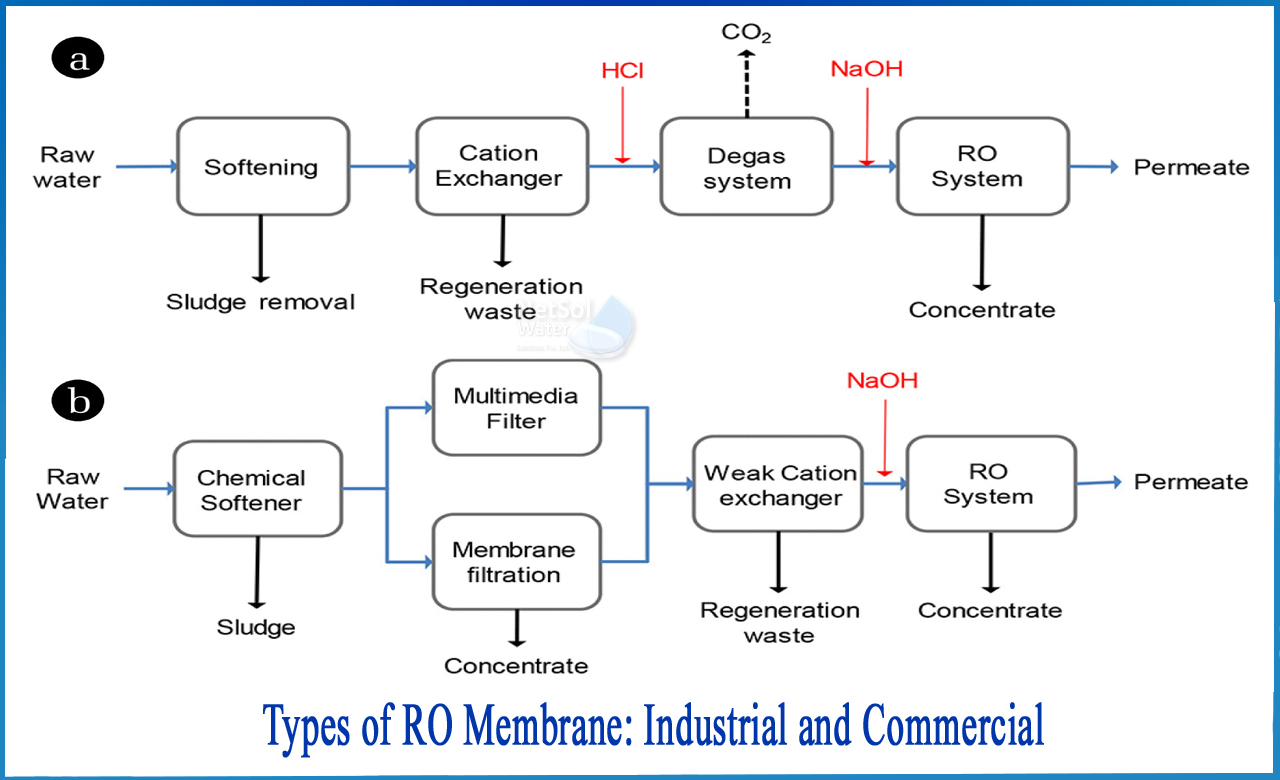
3.REVERSE OSMOSIS
To remove silica, reverse osmosis or other membrane method can be troublesome. Because of the abrasiveness of the particles, silica, which is similar to glass, will create holes in the membrane and reduce its efficacy. Heavy metals corrode membranes, causing them to fail. Membrane systems should be pretreated to remove the elements that cause membrane fouling. Membranes will work ideally without having to be replaced if pretreatment is done prior to membrane treatment.
4.ELECTROCOAGULATION (EC)
A simple, cost-effective, and effective method for removing silica. Silica, heavy metals, and other pollutants can be successfully removed from aqueous solutions using electrocoagulation (EC). Electrocoagulation efficiently eliminates a wide spectrum of pollutants using a single system using clean electricity. The EC renders water constituents "separable." Heavy metals are transformed from ion to oxide forms, enabling for non-hazardous landfill disposal.