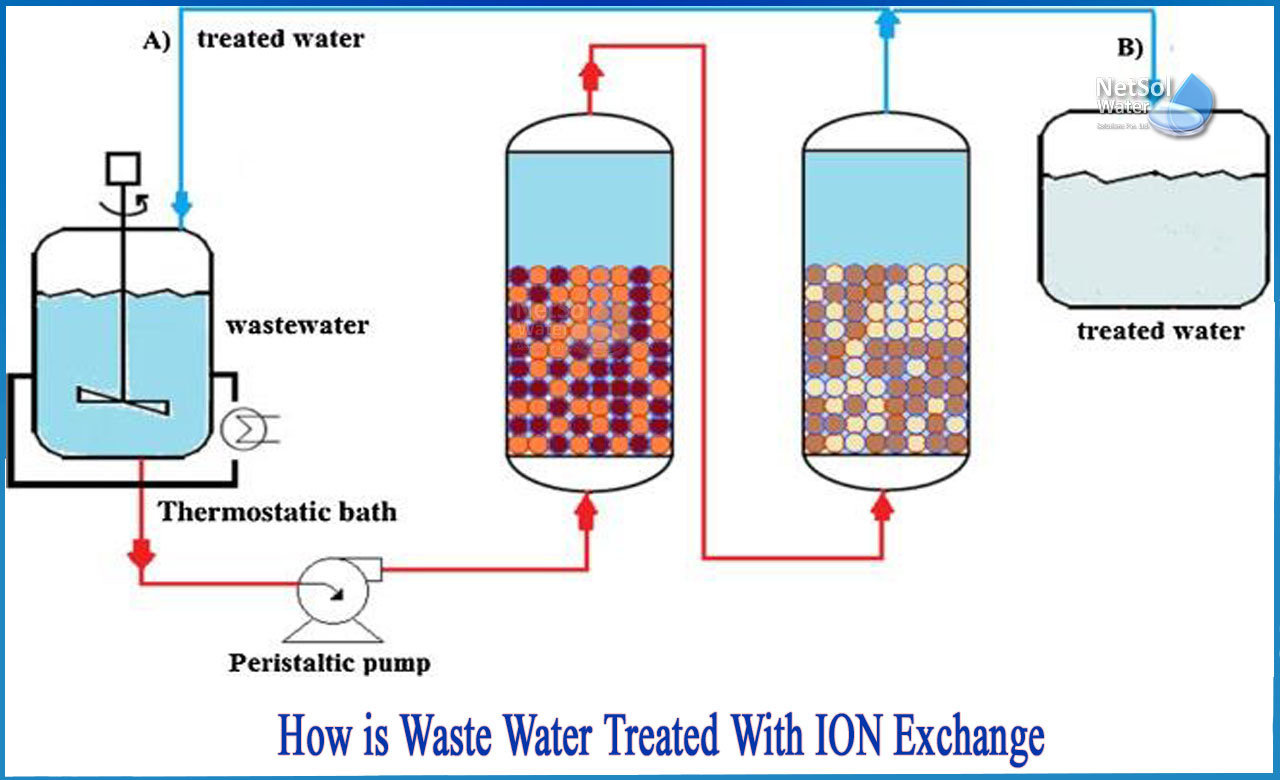
What is the process of ion exchange?
Ion exchange is a procedure in which ions of one species in solution are swapped by ions of a different species bonded to an insoluble resin with a comparable charge. Ion exchange is essentially a sorption process that can also be thought of as a reversible chemical reaction.
Water softening (elimination of "hardness" ions such as Ca2+ and Mg2+) and nitrate separation in advanced wastewater treatment operations are two major applications of ion exchange. These ion exchange resins are either inorganic zeolites that exist naturally or organic resins that are synthesised. Because their properties may be adjusted to specific uses, synthetic organic resins are the most often utilised nowadays.
What are the ion exchange resins made of?
An organic ion exchange resin is made of an organic or inorganic network structure with functional groups attached, that can exchange mobile ions for ions with similar charges in the extracellular environment. The largest amount of exchanges per unit of resin is determined by the amount of mobile ion sites present in each resin.
Cationic ion exchange resins exchange positive ions, while anionic ion exchange resins swap negative ions. Cation exchange resins have acidic functional groups like sulfonic, but anion exchange resins are often characterised as strong acid, weak acid, strong base, or weak base based on the type of the functional group. Similar to soluble acids and bases, the strength of the acidic or basic character is determined by the degree of ionisation of the functional groups. As a result, a resin containing sulfonic acid groups would be a good cation exchange resin.
Resins of Ion Exchange
Ion exchange resins are divided into two types: cation exchangers, which contain positively charged mobile ions available for exchange, and anion exchangers, which have negatively charged exchangeable ions. The same basic organic polymers are used to make anion and cation resins. The ionizable group linked to the hydrocarbon network differs. The chemical behaviour of the resin is determined by this functional group. Strong or weak acid cation exchangers and strong or weak base anion exchangers are the two broad classifications for resins.
Application of Ion exchange in waste water treatment
The elimination of salt at low quantities, as well as the demineralization and softening of water, the retention of specific chemical compounds, and the demineralization of sugar syrups, are all common applications of these systems.
Characteristics of Ion exchange
The following are the properties that govern the ion exchange process and determine its main characteristics at the same time:
1. The resins are selective, meaning they may prefer one ion over another with relative affinity values of 15 or higher.
2. The ion exchange reaction is reversible, meaning it can proceed in both ways.
3. The reaction maintains electroneutrality.
Advantages of treatment of waste water with ion exchange
1. If user is working with relatively low salt concentrations, the equipment is quite adaptable.
2. Resins now offer excellent treatment capabilities and have shown to be compact and cost-effective.
3. Chemically, resins are exceedingly stable, long-lasting, and simple to regenerate.
4. Automatization and adaption to unique settings are both quite simple
Conclusion
In treating waste water, ion exchange technology can be used. As being the best manufacturer of waste water treatment plants in India, we, Netsol Water, assure all our customers that they will have access to the best waste water technology with all needs and services.
Netsol Water is Greater Noida-based leading water & wastewater treatment plant manufacturer. We are industry's most demanding company based on client review and work quality. We are known as best commercial RO plant manufacturers, industrial RO plant manufacturer, sewage treatment plant manufacturer, Water Softener Plant Manufacturers and effluent treatment plant manufacturers. Apart from this 24x7 customer support is our USP. Call on +91-9650608473, or write us at enquiry@netsolwater.com for any support, inquiry or product-purchase related query.