What is Degasification?
Degasification is the process of removing dissolved gases from liquids. It is based on "Henry's Law," or to be more precise, the "proportionality factor is termed as the Henry's law constant," which was created by William Henry in the early 1800s.
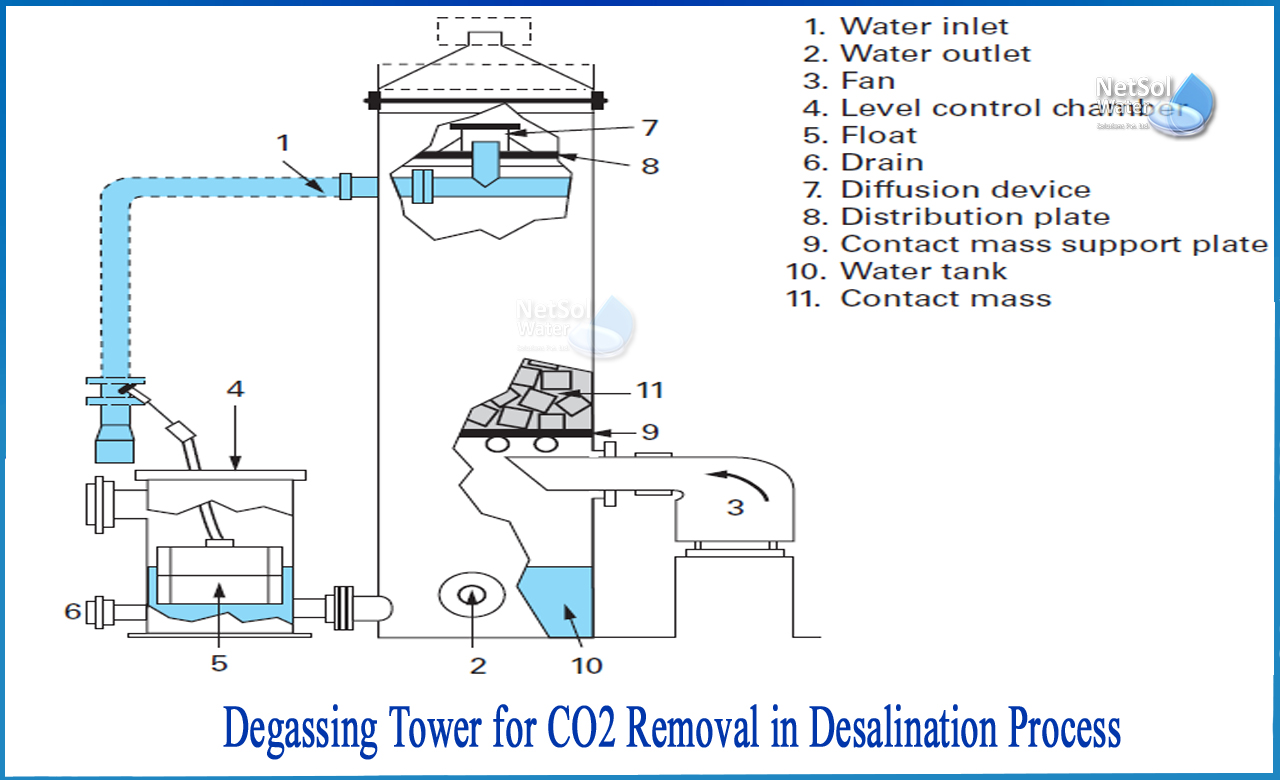
According to Henry's Law, the amount of dissolved gas in a gas is proportional to its partial pressure. The packed vertical tower known as a degasifier or decarbonator is the most effective procedure of degasification. Water arrives at the top of the tower and flows down a media bed by gravity. The media bed acts to repeatedly reshape the water, revealing any dissolved gas molecules to the water droplet's surface. Air is introduced in a cross current approach, either by force or by induction, that crosses over the water droplets and "strips" the gas molecules out of the water at the same time that the water is moving down the inside of the tower. The stripped gases are then expelled from the tower through the exhaust at the top.
This is the "beginner's approach to water degasification."
Most Common degassing processes are:
· Thermal Degassing
1. Pressure Degassing
2. Vacuum Degassing
· Membrane Degassing
· Chemical Degassing
What is the reason to perform degasification?
Degasification is required in the manufacture of chemicals, medications, and even cosmetics, but the most common demand is for water. When water is used to make steam, dissolved gases such as CO2 (carbon dioxide) must be eliminated before the water enters the boiler to avoid corrosion and scaling. When CO2 is left in water, it can develop carbonic acids, which can lead to corrosion and degrade the performance and longevity of the steam-generating equipment. When water is processed for human consumption, it is frequently degasified to eliminate CO2 and other dissolved gases including such hydrogen sulphide (H2S), also known as "rotten egg gas" by customers.
Is degasification a cost-effective method?
It is a technique for eliminating carbon dioxide (CO2) from water, which raises the pH of the water by eliminating carbon dioxide (CO2) gases. The pH of water is frequently lowered prior to reverse osmosis to help prevent membrane scaling, therefore the pH of the water may still be in the acidic range after it exits the membrane filtration process. If degasification was not used to reduce the pH, more chemicals, such as caustic (a base), would be required to return the pH to a neutral level of 7. When compared to the operational costs of a normal water treatment facility, degasification with a degasification tower is significantly more cost effective than chemical use.
Conclusion
Netsol Water is a leading manufacturer in India, of all water treatment plants including degasification towers, and their systems have been consistently improved to increase efficiency and minimise running costs. The distribution system, for example, is an important component in starting the degasification process when water first reaches the treatment tower. A degasification tower's performance is also influenced by the type of media used and the air vane flow mechanism. To better assist the water professional, Netsol Water can customise one of the first "smart" degasification towers in the market.
Netsol Water is Greater Noida-based leading water & wastewater treatment plant manufacturer. We are industry's most demanding company based on client review and work quality. We are known as best commercial RO plant manufacturers, industrial RO plant manufacturer, sewage treatment plant manufacturer, Water Softener Plant Manufacturers and effluent treatment plant manufacturers. Apart from this 24x7 customer support is our USP. Call on +91-9650608473, or write us at enquiry@netsolwater.com for any support, inquiry or product-purchase related query.