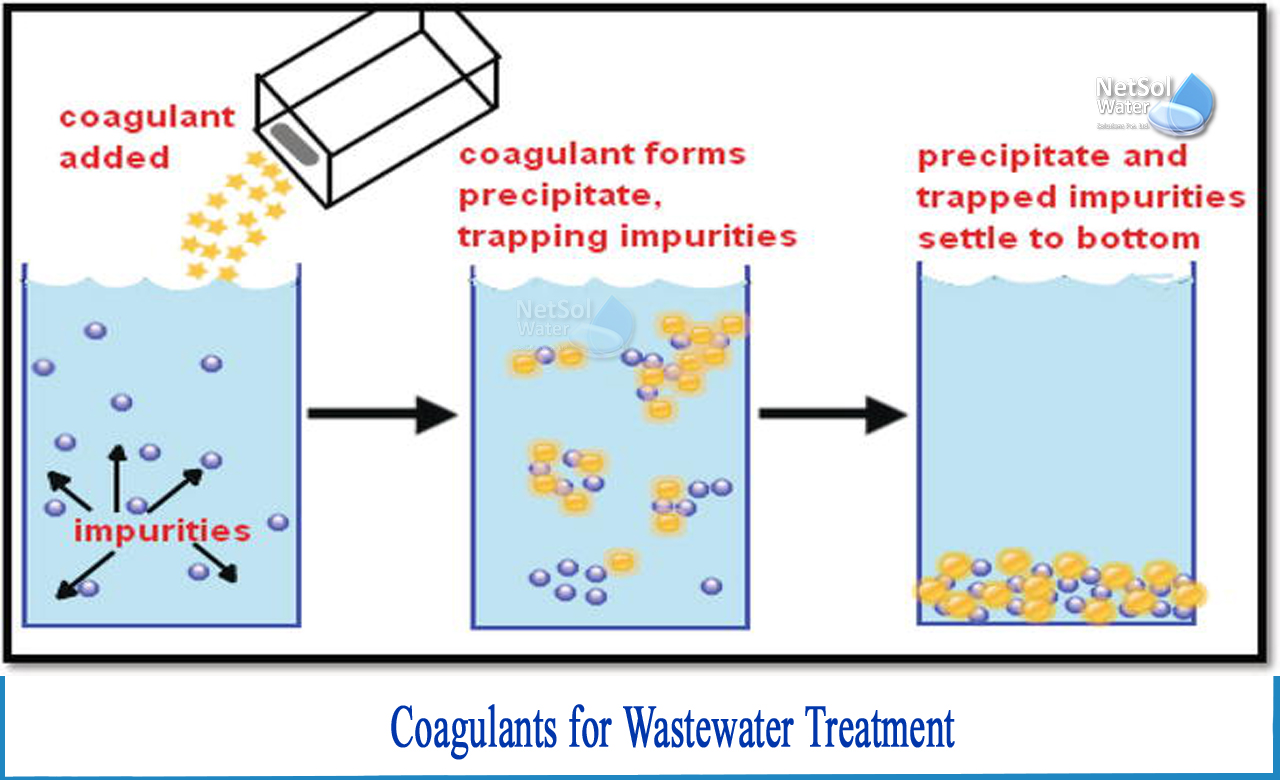
What is coagulation?
Curdling and clotting are synonyms for coagulation, which is the process of a liquid converting to a solid form. It's a chemical reaction in which a liquid's chemical characteristics change, causing particles to clump together and create solid matter. Coagulation, like flocculants, has been used to filter water for over 4,000 years and is still one of the most common ways used today.
What are Coagulants?
Compounds that enhance the coagulation of liquids into solids are known as coagulant chemicals.
The two types of chemical coagulants for water treatment are organic and inorganic.
1: Organic Coagulant: Generally, these are used to separate solids from liquids and to produce sludge. Polyamines (e.g. poly-diallyl dimethylammonium chloride), which are the most extensively used organic coagulants, are the first type of organic water treatment chemicals involved. They function by simply neutralising the charges of particles so that they can join together, and they are most successful at treating wastewater and high turbidity raw water. Melamine formaldehyde and tannins are the second category of organic wastewater chemicals, and they are utilised to coagulate colloidal particles in water. These are particularly well-suited to the treatment of hazardous sludge because they effectively absorb organic contaminants such as oil and grease.
2: Inorganic Coagulant:Inorganic coagulants are frequently more cost-effective than their organic equivalents, and they can be used in a wide range of water treatment operations. However, because they are acidic in nature, they must be handled and stored with caution. Inorganic coagulants are especially effective in treating low-turbidity raw water, and they can be used to treat this type of water when organic coagulants fail. The majority of inorganic coagulants are made of aluminium or iron. Other chemicals used to treat wastewater include aluminium chloride, polyaluminium chloride, aluminium chlorohydrate, ferric and ferrous sulphate, and ferric chloride.
Why are coagulants used in waste treatment?
Coagulants are used to remove harmful pollutants from water, including pathogens and organic waste as well as toxic elements like chemical phosphorous and chloride and inorganics. They are frequently used as the initial stage in a pollution reduction plan for developing countries, particularly those with rapidly growing urban areas and no centralised wastewater treatment. Coagulants are utilised in medical applications to cause blood clotting, in addition to being chemicals used in water treatment plants. They're also employed in the production of pulp, paper, and textiles, as well as in oil, gas, and mining operations.
Working of Coagulants
Coagulants are positively charged chemicals that act by neutralising the electrical charge of particles, causing the magnetic forces that maintain colloids apart to destabilise. When inorganic coagulants are introduced to wastewater and the cationic metal ion neutralises the colloid particles, iron or aluminium precipitates form, which absorb pollutants and purify the water. The sweep-floc mechanism is used in this process, which, while effective, adds to the overall volume of sludge that needs to be treated and removed. The procedure for organic chemical coagulants is similar, except that instead of metal, the positive charge is usually provided by an amine (NH4+) group linked to the coagulant molecule.
Conclusion
Coagulants and flocculants, which have been used to filter water for thousands of years, are still important components of current water treatment operations. Wastewater treatment must always be performed by skilled professionals in licenced facilities due to the hazardous nature of both the chemicals and the labour involved.
Contact our knowledgeable team if you have any queries concerning coagulants, flocculants, or other wastewater treatment products and procedures.
Netsol Water is Greater Noida-based leading water & wastewater treatment plant manufacturer. We are industry's most demanding company based on client review and work quality. We are known as best commercial RO plant manufacturers, industrial RO plant manufacturer, sewage treatment plant manufacturer, Water Softener Plant Manufacturers and effluent treatment plant manufacturers. Apart from this 24x7 customer support is our USP. Call on +91-9650608473, or write us at enquiry@netsolwater.com for any support, inquiry or product-purchase related query.