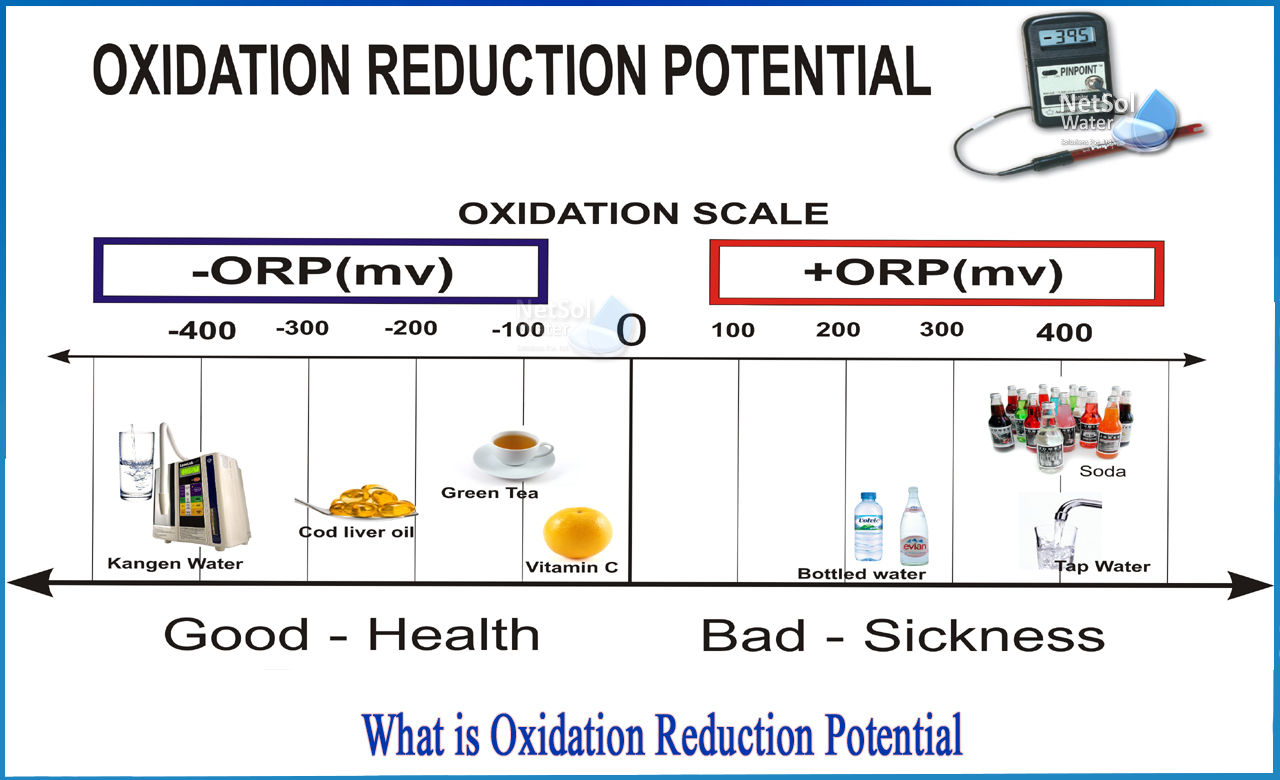
What do you mean by oxidation reduction potential?
The oxidation-reduction potential (ORP) of a lake or river measures its ability to cleanse itself or break down waste products such as contaminants and dead plants and animals. When the ORP value is increased, the water contains a lot of oxygen. It means that bacteria that decompose dead tissue and contaminants will be able to work more effectively.
The higher the ORP value, in general, the healthier the lake or river. However, even in healthy lakes and rivers, as you get closer to the bottom sediments, there is less oxygen (and thus lower ORP values). This is due to the fact that many bacteria are working hard in the sediments to decompose dead tissue, and they consume a large amount of the available oxygen.
In fact, oxygen vanishes quickly in the bottom mud (often within a centimetre or two), and ORP falls rapidly.ORP is measured in relation to dissolved oxygen because it can provide scientists with additional information about the water's quality and level of pollution, if present. There are some other elements that, in terms of chemistry, can function similarly to oxygen and contribute to improved ORP.
Why does oxidation reduction potential matter?
ORP is affected by the amount of dissolved oxygen in the water as well as the presence of other elements that function similarly to oxygen. Though not technically correct, oxygen and other elements that contribute to increased ORP effectively help 'eat' things in the water that we don't want – such as contaminants and dead tissues.
When the ORP is low, the dissolved oxygen level drops, the toxicity of certain metals and contaminants rises, and there is a lot of dead and decaying material in the water that cannot be passed or broken down. This is clearly not a good environment for fish or bugs. ORP should be high in healthy waters, ranging between 300 and 500 millivolts.
How do we measure oxidation reduction potential?
Using an ORP sensor, ORP is measured directly in the lake or river water that you are investigating. ORP is evaluated in millivolts (mV), and the higher the ORP reading, the more oxygen present in the water. ORP can be either greater than or less than zero.
Application of ORP:
Water disinfection is one of the most important applications of ORP. To kill bacteria and other microbes and prevent their growth in water supply lines, municipal drinking water supplies, for example, use strong oxidizers such as chlorine. Because higher ORP values are associated with higher disinfectant concentrations, ORP is used to monitor and control disinfectant levels in water supplies.
Disinfectants are used in swimming pools and spas to kill microbes that can transmit diseases. Disinfectants are also used to prevent algae growth in outdoor swimming pools and cooling towers.
ORP can also be used in industrial processes to monitor and control many oxidation-reduction reactions. ORP is used in wastewater treatment to determine the types of microbial processes that are occurring and to assist operators in managing the treatment system by promoting or inhibiting specific reactions.ORP measurements can be viewed as an extension of the dissolved oxygen (DO) scale in other environmental applications. DO metres can detect a variety of aerobic conditions, but they cannot detect how reducing an anaerobic environment is.
In contrast, the ORP scale encompasses a wide range of reducing conditions. As a result, ORP can provide information about the chemistry of anaerobic environments, such as the types of microbial processes in sediments or pollutant reactions in contaminated aquifers.ORP can also be used in combination with membrane DO sensors to identify conditions where DO measurements may be incorrect.
Netsol Water is Greater Noida-based leading water & wastewater treatment plant manufacturer. We are industry's most demanding company based on client review and work quality. We are known as best commercial RO plant manufacturers, industrial RO plant manufacturer, sewage treatment plant manufacturer, Water Softener Plant Manufacturers and effluent treatment plant manufacturers. Apart from this 24x7 customer support is our USP. Call on +91-9650608473, or write us at enquiry@netsolwater.com for any support, inquiry or product-purchase related query.