What are the various methods of measuring Dissolved oxygen?
A simple chemical analysis method (titration method), an electrochemical analysis method (diaphragm electrode method), and a photochemical analysis method may all be used to detect dissolved oxygen levels (fluorescence method).
A: Titration Procedure
1: The Winkler Method
When a manganese peroxide solution and a sodium hydroxide solution are combined with sample water, a precipitate of manganese hydroxide (II) is formed. This manganese hydroxide (II) precipitate interacts with dissolved oxygen in the water and is oxidized in proportion to the quantity of DO, leaving a brown precipitate.
If DO is missing:
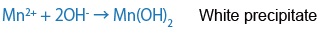
If it reacts with DO:

If you dissolve this brown precipitate in an acid containing iodine ions (I-), iodine (I2) will be liberated in proportion to the amount of DO. The amount of liberated iodine is then determined by titration with sodium thiosulfate.
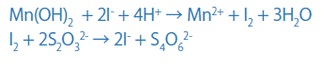
Modified Winkler’s Method Using Sodium Azide
The goal of this approach is to increase the accuracy of DO measurement using Winkler's method. The I2 that remains after the titration with sodium thiosulfate in the last procedure of Winkler's technique, is titrated with a starch solution again in this approach.
2: Miller's technique is another titration method.
B: Diaphragm Electrode Technique
Dissolved oxygen levels are commonly measured using the diaphragm electrode technique. This technique uses electrodes to measure the amount of oxygen passing through a highly oxygen permeable diaphragm. The galvanic electrode method and the polarographic method are two ways for determining dissolved oxygen levels using electrodes.
C: Galvanic Dissolved Oxygen Sensors
Galvanic is the electrochemical dissolved oxygen sensor. The electrodes of a galvanic dissolved oxygen sensor are made of different metals.
Based on their activity series, metals have varying electropotentials (how readily they give or accept electrons). When different metals are introduced in an electrolyte solution, the potential between them leads them to self-polarize. Because of its self-polarization, a galvanic DO sensor does not require any warm-up time. To eliminate oxygen without an external applied potential, the potential difference between the anode and the cathode must be at least 0.5 volts.
-Zn/Pb – Zinc or lead anode
-NaCl and H2O – Sodium chloride solution
-Ag – Silver cathode *inert electrode, does not react*
Zinc Anode Reaction and Oxidation
2Zn —-> 2Zn2+ + 4e–
Silver Cathode Reaction and Oxygen Reduction
The Ag cathode is inert and only passes electron without participating in the reaction
O2 + 4e– + 2H2O —-> 4OH–
4OH– + 2Zn2+ —-> 2Zn(OH)2
Overall Reaction
O2 + 2H2O + 2Zn —-> 2 Zn(OH)2
Because the cathode is an inert electrode, it is left out of the equation, as it is in the polarographic dissolved oxygen sensor reaction. The silver cathode receives electrons from the anode and transfers them to oxygen molecules. This transaction takes place at the cathode's surface. The current generated by oxygen reduction is proportional to the partial pressure of oxygen in the water sample.
D: Sensors for Pulsed Polarographic Dissolved Oxygen
When detecting dissolved oxygen, pulsing polarographic dissolved oxygen sensors eliminate the need to mix the sample. A steady-state polarographic DO sensor is similar to a rapid-pulse DO sensor in that both use a gold cathode and a silver anode. Both steady-state and rapid-pulse sensors detect dissolved oxygen by polarizing the electrodes with a constant voltage.
D: Fluorescence method
The fluorescence method for monitoring DO levels is a relatively new technology that has lately gained popularity for usage in field settings since it does not necessitate the change of the internal solution. The quenching phenomena of oxygen is used in this approach to calculate the amount of DO. The fluorescence of a fluorescent material stimulated by blue light-emitting diode (LED) light is suppressed by oxygen passing through a DO permeable layer. The stronger the quenching event and the less fluorescence the detector (light-receiving diodes) detects, the higher the DO level.
What do we offer?
Netsol Water has a significant presence in India and is well positioned to help stimulate a national effort to address the country's water crisis. We want to deliver knowledge regarding water and wastewater to every nook and corner, so that more and more water is saved by every human being. We are the large water and wastewater treatment company in India that manufactures WTPs, WWTPs, STPs, ETPs, Industrial & Commercial RO Plants, among other services. We give our 100 percent in converting the wastewater into best possible treated water, as we are committed to do so.
Netsol Water is Greater Noida-based leading water & wastewater treatment plant manufacturer. We are industry's most demanding company based on client review and work quality. We are known as best commercial RO plant manufacturers, industrial RO plant manufacturer, sewage treatment plant manufacturer, Water Softener Plant Manufacturers and effluent treatment plant manufacturers. Apart from this 24x7 customer support is our USP. Call on +91-9650608473, or write us at enquiry@netsolwater.com for any support, inquiry or product-purchase related query.



