What is demineralization process of water softening?
Removal of contaminants or impurities like calcium, magnesium, silica and iron which are prime causes for scale formation is known as water softening or demineralization. Some of the conventional methods to do so are:
- Lime Softening.
- Sodium Zeolite Softening.
- Reverse Osmosis.
- Electro dialysis.
- Ion Exchange method etc.
The treatment method employed mainly depends upon the raw water quality, demand and purity requirements. It must be also noted that the process also depends upon the cost involved- budget.
QUICK LIME AND CLARIFIERS
When slaked lime or quick lime is added to raw water having hardness, it reacts with the minerals present (calcium and magnesium) to form solid precipitates. This process usually takes place in a clarifier. The precipitate is in solid state and the reaction takes place in rapid mix zone. The solidified form is removed from the bottom of the tank which allows the treated water to flow over. The effluent is now treated water and is free from hardness. This water is suitable for boilers as well.
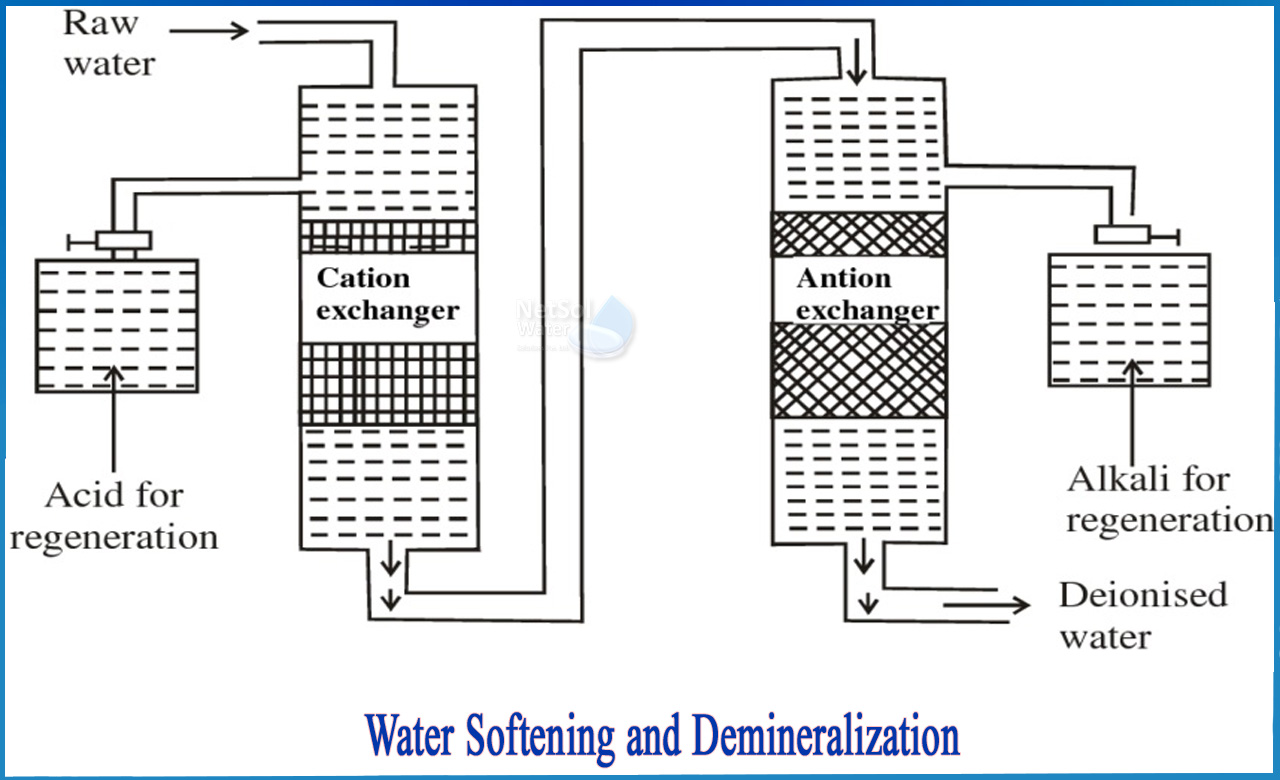
ION EXCHANGE
Ion exchange as the name suggests means that exchange occurs in the process. A process that exchanges one charged particle with another. The impurities that occur in water are charged particles, making the process vital in boiler treatment process.The process takes place in the closed container which is partially filled with charged resin. The resin is insoluble, material made up of plastic and is capable of substitution for another ion. There are two types of ions involved in the process and each one is capable of changing the other. The types are as:
· Cation = positively charged Ions
· Anion = negatively charged Ions
In order to ascertain quality water, elaborate systems are usually employed but the basic working principle remains same. Ion exchange resins have one serious disadvantage that they possess limited capacity and finally become exhausted. There are enough processes which are employed for regeneration but the regeneration occurs with salt brine which adds up the step to mix it with fresh water such that no salt enters the process.
DEALKALIZERS
The alkalinity of softened water through ion exchange process can be reduced with the help of dealkalizers. The water is made to pass through anionic exchange resin where carbonate, bicarbonate and sulphate ions are exchanged for chloride ions. The regeneration takes place with the help of sodium chloride and softened water. Small quantity of caustic may add up during regeneration cycle. This happens in order to increase capacity and increase in hydrogen ion concentration.
Prevention of carbon dioxide generation inside the boiler is the primary benefit of dealkalized water. Carbon dioxide leaves the boiler with the steam and can form carbonic acid in the condensate, leading to the primary cause of condensate system corrosion.
OTHER TECHNOLOGIES
Other technologies are employed to remove undesirable impurities from the water supply, including reverse osmosis, electro dialysis, which is achieved by reversing the current. These are also called as membrane processes. Reverse osmosis uses semipermeable membranes that let water through but block the passage of salts because the membrane hinders the passage of salts. In the case of electro dialysis, the salts dissolved in the water are forced to move through cation-selective and anion-selective membranes, removing the ion concentration and thus helps to clear the hardness.
Netsol Water is Greater Noida-based leading water & wastewater treatment plant manufacturer. We are industry's most demanding company based on client review and work quality. We are known as best commercial RO plant manufacturer, industrial RO plant manufacturer, sewage treatment plant manufacturer, Water Softener Plant Manufacturers and effluent treatment plant manufacturers. Apart from this 24x7 customer support is our USP. Call on +91-9650608473, or write us at enquiry@netsolwater.com for any support, inquiry or product-purchase related query.