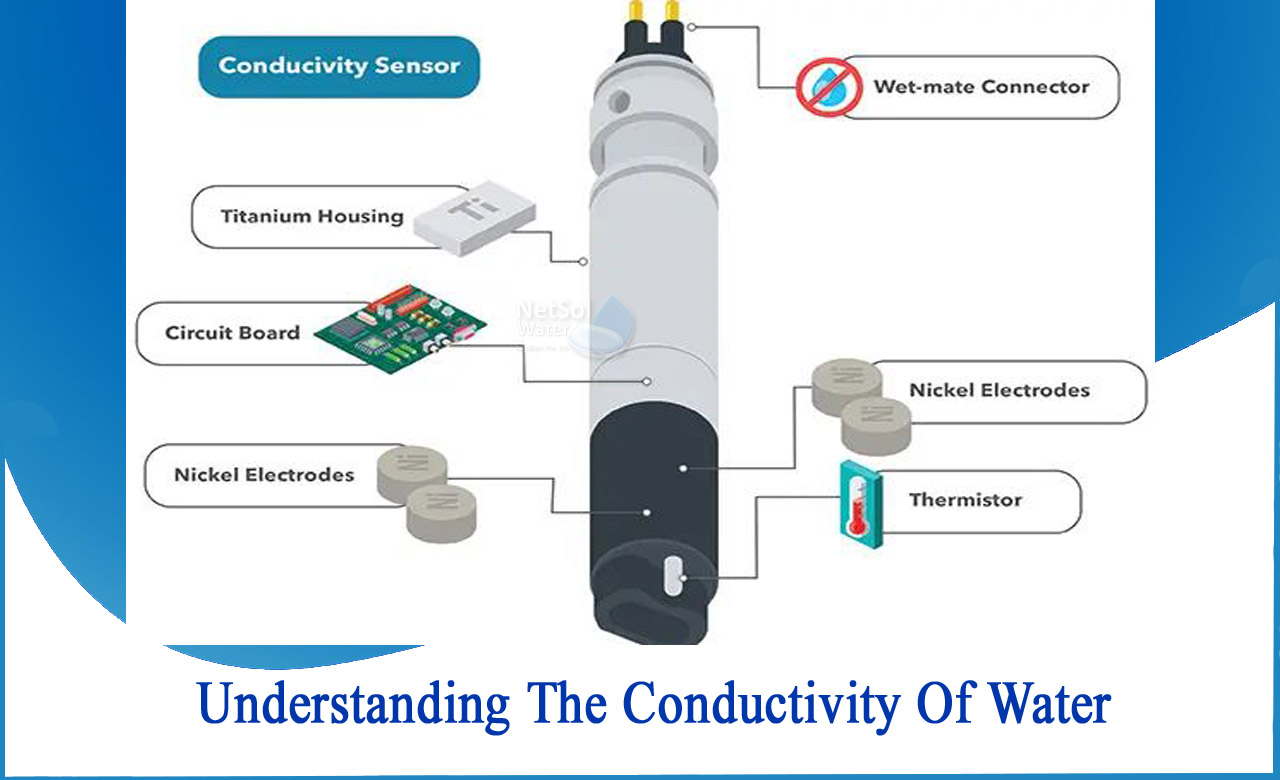
WHAT IS CONDUCTIVITY?
The ability of water to conduct an electrical current is measured by its conductivity. Conductivity rises as salinity rises because dissolved salts and other inorganic compounds conduct electrical current. Oil and other organic molecules do not transmit electrical current well and so have a low conductivity in water. Temperature has an impact on conductivity: the warmer the water, the higher the conductivity.
When different chemicals and salts dissolve in water, they form negatively charged and positively charged ions. Potassium, magnesium, and sodium are examples of positively charged ions that can alter water. Carbonate, chloride, and sulphate, on the other hand, are negatively charged ions. Scale accumulation can occur when a chemical like sulphate is present in higher concentrations, causing damage to boilers and other industrial equipment.Measuring the electrical conductivity of water is a fairly simple and affordable method of determining how many ions are present in the water for environmental and industrial applications. If necessary, the water can be treated after these measurements have been taken.
The conductivity of water can be used as a broad indicator of its quality. Each water body has a somewhat stable conductivity range that can be used as a baseline for comparison with regular conductivity measurements once established. Significant variations in conductivity could indicate that pollution has reached the aquatic resource from a discharge or another source.
Significant variations in conductivity (typically rises) may indicate that a discharge or other source of disturbance has lowered the relative state or health of the water body and its biota. Human disturbance generally increases the amount of dissolved solids entering waterways, resulting in higher conductivity. Other indications may be hindered or altered in water basins with increased conductivity.
HOW IS WATER CONDUCTIVITY MEASURED?
1- One can measure water conductivity with a variety of equipment’s available in the market, including:
2- HandheldTesters are meters that may be dropped into water to measure a variety of parameters, including conductivity, salinity, temperature, and pH levels.
3- Portables are water quality monitoring meters that are ideal for use in the field. These gadgets are used by environmental researchers because they are easy to read in outdoor settings.
4- In a laboratory setting, benchtopmeters can be used to detect conductivity. They're often larger than portable testers and can provide more accurate readings.
5- Inlinemeters are permanently mounted at specific positions along a production line to measure water quality characteristics in real time, including conductivity.
6- Electrodes are used in order to examine the characteristics of liquids, electrodes are also employed to carry electrical currents (e.g., how conductive they are).
WHY IS WATER CONDUCTIVITY IMPORTANT?
Water quality is critical to many sectors and the general well-being of the populace. Conductivity, which indicates how many pollutants are present in the water and have the potential to transmit electricity, can be used to indirectly quantify water quality.
Baseline readings can be maintained in check to preserve high-quality water with healthy conductivity levels because conductivity is commonly measured in micro-Siemens per centimeter (S/cm) or 1000 times that, milli-Siemens per centimeter (mS/cm).
1- Researchers and scientists need to know the exact composition of the water they're experimenting with in order to get reliable, repeatable results, hence water conductivity must be measured in lab settings. Purified water is widely used in the pharmaceutical, semiconductor, and solar industries for this reason.
2- Water conductivity must be assessed in the food and beverage business to ensure that high-quality end products are produced effectively, improving profitability and reducing hazards.
3- Water conductivity is another something that aquaculture and aquarium businesses need to be aware of. For example, freshwater and saltwater fish may require different degrees of water conductivity to thrive. Aquarium and aquaculture businesses can track conductivity levels to see if the water in their tanks is changing. They can take proactive measures to restore the water's equilibrium now that they have this information.
4- Along with this, Conductivity affects salinity, which is the total saltiness of a body of water, in addition to the health of ocean water. The goal of salinity measurements is to determine how much dissolved salts are present in water.
5- When electrolytes dissolve, they produce ionic particles, which have a negative and positive charge. Because salinity is difficult to detect directly, conductivity measurements are used to determine the salinity of the water. Carbonates, calcium, sodium, and magnesium are just a few of the numerous types of soluble salts that can raise the salinity of water.