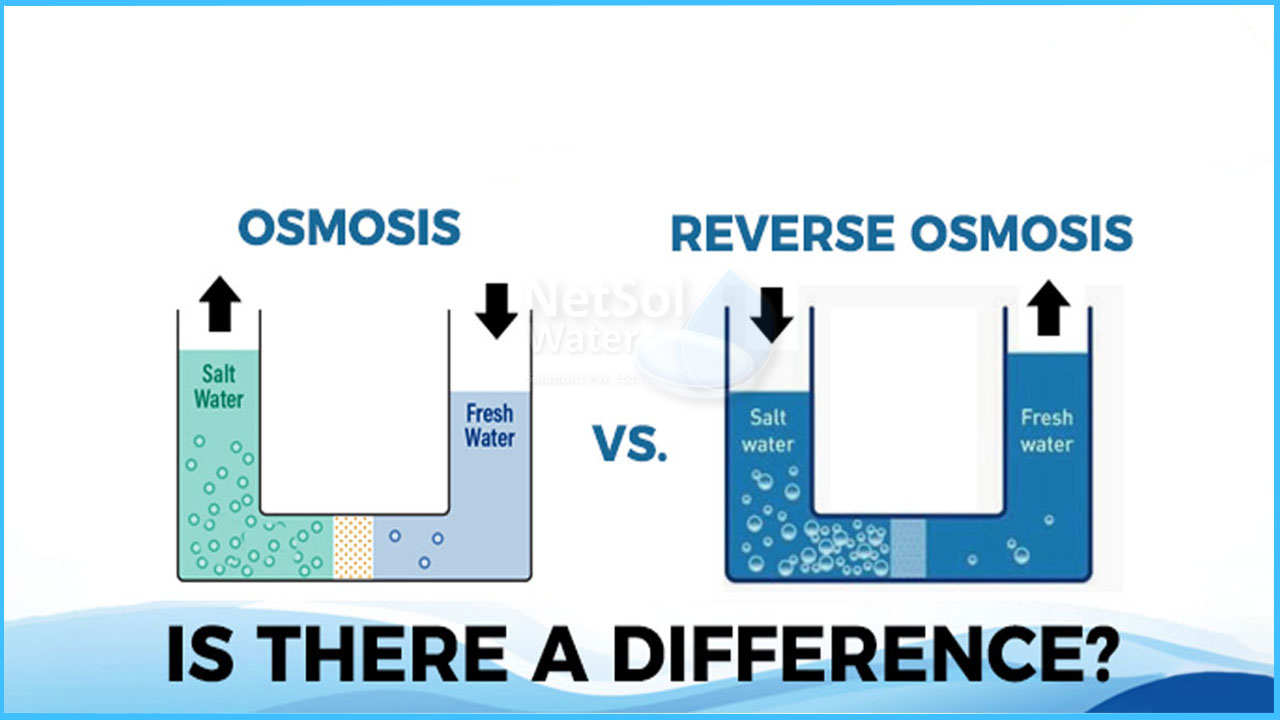
The main distinction between osmosis and reverse osmosis is that osmosis is a natural process in which water molecules pass along a concentration gradient, whereas reverse osmosis is a water purification procedure in which water molecules pass across a semi-permeable membrane against the concentration gradient.
What is Osmosis?
Osmosis is a natural process that occurs in all living things on a regular basis. It is the passage of water molecules through a semi-permeable membrane from a higher water potential area to a lower water potential area. Osmosis does not require energy because it occurs along a concentration gradient. As a result, it is a passive procedure. In both plant and animal cells, osmosis is the primary process that allows water to pass through the cell membrane. Because the cell membrane is selectively permeable, only some molecules can pass through it. Water molecules and solvent molecules can only enter and exit the cell by osmosis, balancing the solute content inside and outside the cell.
What is Reverse Osmosis?
The process of reverse osmosis is used in the desalination and purifying of water. It's osmosis in the other direction, as the name implies. Water is pushed through a semi-permeable membrane against the concentration gradient by applying a pressure larger than the natural osmotic pressure. As a result, water molecules migrate from a low water potential to a higher water potential across a reverse osmosis membrane. Dissolved salts, organics, microorganisms, and pyrogens, for example, will not flow through the barrier. As a result, reverse osmosis makes it easier to filter water in water purification procedures.
Osmosis vs. reverse osmosis?
The movement of water molecules through a semi-permeable membrane are referred to as osmosis and reverse osmosis. Osmosis, on the other hand, occurs passively along the concentration gradient, whereas reverse osmosis occurs actively with the expenditure of energy. This is the primary distinction between osmosis and reverse osmosis. Furthermore, natural osmotic pressure affects the process in osmosis, whereas a pressure stronger than natural osmotic pressure is provided in reverse osmosis to transfer water molecules against the concentration gradient. As a result, there is a fundamental distinction between osmosis and reverse osmosis.
Another distinction between osmosis and reverse osmosis is that, unlike osmosis, reverse osmosis necessitates the use of energy to maintain pressure. If we talk about direction, osmosis occurs from high water concentration to low water concentration while in case of reverse osmosis, the direction is opposite, it occurs against the concentration gradient. All these processes acting differently make the difference clear between osmosis and reverse osmosis.