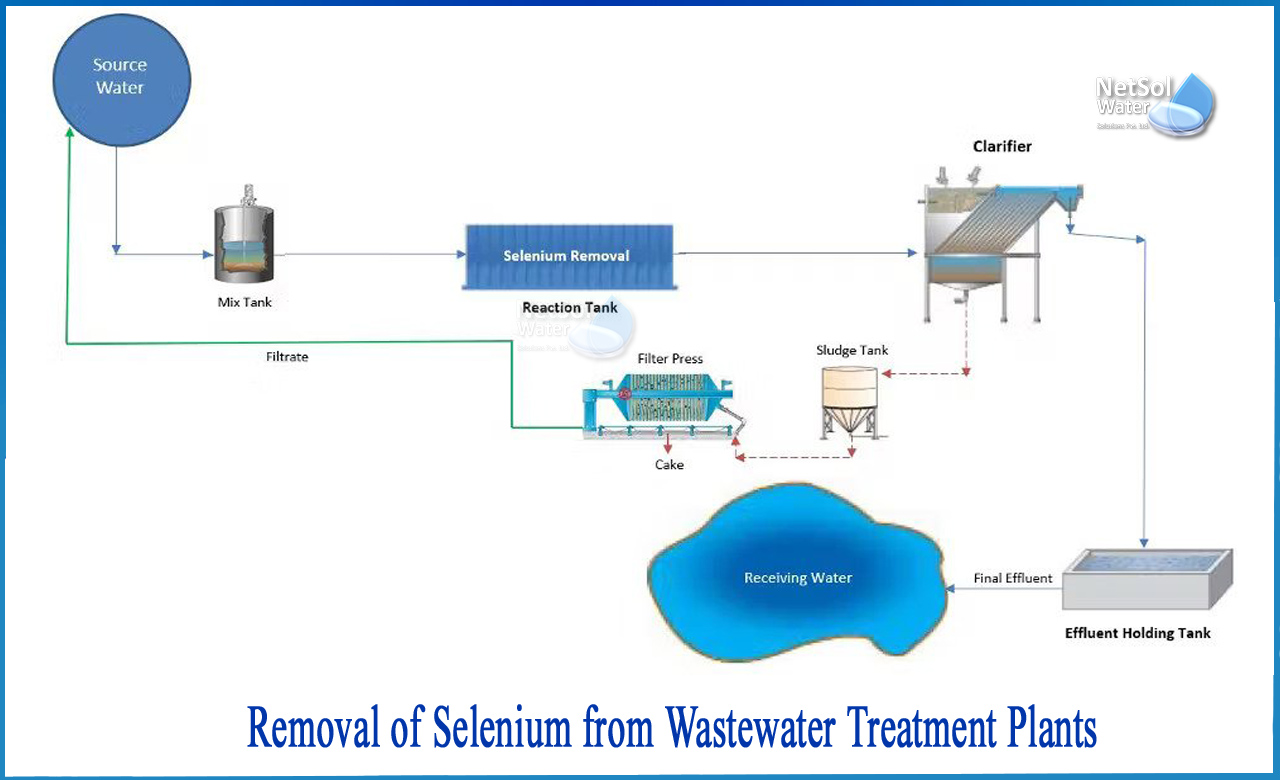
How to remove selenium from Wastewater treatment plants?
High concentrations of selenium salts are poisonous, yet minimal levels are required for cellular activity in many species, including animals. Many multivitamins and other nutritional supplements, including infant formula, include selenium.
The by-products of coal combustion in power plants, known as coal combustion residuals (CCRs), contain pollutants such as selenium, mercury, cadmium, and arsenic, which have been linked to a variety of negative health impacts.
These pollutants can be found in large amounts in coal-fired power plant wastewater. The rules prohibit the storage of additional CCR waste in surface ponds and encourage coal plants to dewater and close ponds as soon as possible. The risk of toxins leaking into groundwater, blowing into the air as dust, or being discharged into surface water due to catastrophic failure of coal ash impoundments is a problem with CCR storage in ash ponds. Growing legislation and environmental concerns make it critical to select an efficient and long-term treatment strategy for coal ash ponds.
Using a Mobile Solution to Remove Selenium from Coal Ash Ponds
Selenium treatment methods frequently rely on technology that isn't typically found in a regular wastewater treatment system and necessitates extra, specialised handling. However, selenium may be removed from wastewater by temporarily deploying mobile equipment at the ash pond to give a cost-effective treatment method that employs standard equipment.
The equipment required for selenium treatment is not included in most wastewater treatment systems, and adding the extra pieces adds to the complexity. The following are examples of specialised treatment processes:
· Reverse osmosis or ion exchange, both with following brine treatment, and
· Biological treatment specially designed to target selenium after filtration
Selenium can be present in two different forms in water:
1. Selenite, Se4+, is a reduced form of selenium
2. Selenate, Se6+, is oxidised selenium
Small dosages of oxidant are not adequate to oxidise the selenium. Approaching selenium removal in ash ponds through the arsenic treatment process using a standard water treatment system is a cost-effective and feasible solution. Because the same chemical method that is used to treat arsenic can also be used to treat selenium, the selenium can be lowered with minimal additional equipment.
Conclusion
Netsol's selenium treatment solution employs an arsenic-targeting method that follows a standard treatment scheme. Coagulation and oxidation, filtration, sedimentation, and filtration are all procedures used in some form or another in most wastewater treatment systems to treat high levels of selenium.
1: Selenium species are rarely oxidised on purpose since the selenate form is difficult to eliminate. Reduced species, such as selenocyanate found in stripped sour water used in oil refining, must be oxidised to selenite or selenate before biological therapy may be used. You can use a typical oxidant like chlorine, hydrogen peroxide, or potassium permanganate. Biological therapy or the chemical treatment techniques are usually used to achieve reduction.
2: Iron co-precipitation is a technique that involves precipitating significant volumes of ferric oxide/hydroxide while also collecting low concentrations of other pollutants within the ferric compounds. Selenite is successfully eliminated by iron co-precipitation, but only a small amount of selenate is eliminated. Because it generates huge amounts of sludge, this method is commonly utilised at mine sites and typically necessitates clarifying and filtration downstream of the co-precipitation process.
3: Selenate/selenite ions are exchanged for like-charged ions by employing selenium-specific speciality resins. If competing anions such as nitrate and sulphate are not present or are eliminated first, the resin capacity for selenium can be improved. When the resin capacity is reached, it must be regenerated. After then, the concentrated regenerant must be disposed of or processed further.
Netsol Water is Greater Noida-based leading water & wastewater treatment plant manufacturer. We are industry's most demanding company based on client review and work quality. We are known as best commercial RO plant manufacturers, industrial RO plant manufacturer, sewage treatment plant manufacturer, Water Softener Plant Manufacturers and effluent treatment plant manufacturers. Apart from this 24x7 customer support is our USP. Call on +91-9650608473, or write us at enquiry@netsolwater.com for any support, inquiry or product-purchase related query.