What are phenols?
Phenol is an organic compound that is also known as carbolic acid. Phenols are commonly found in household products such as disinfectants. They are also used in industry for cleaning and the production of certain pesticides and pharmaceuticals. As a result, phenolic compounds are frequently found in industrial effluent.
Phenols are one of the most important chemicals found in modern manufactured goods. It is widely used in the specialty chemicals industry and can be found in everything from aspirin to analysis kits, pharmaceuticals, and paint.
The technology required
Because primary and secondary processes cannot remove phenol from water, tertiary technology is required. Furthermore, phenol contamination can wreak havoc on biological treatment processes, rendering them ineffective. This means that you must remove the pollutant before it enters the biological treatment stage. Because of their noxious nature, phenolic compounds are prohibited from harming aquatic ecosystems or entering drinking water sources.
It is true that without it, modern life as we know it would most likely cease to exist. It truly is the foundation of modern chemistry, allowing talented chemists to create increasingly effective products.
Phenols are extremely useful 'commodity' chemicals that occur naturally in crude oil and are commercially produced as a by-product of the cracking process in oil refining. However, in concentration, it is extremely dangerous and must be handled with extreme caution.
Why is it necessary to remove phenols from wastewater?
Phenols are similar to alcohols in structure, but because they form stronger hydrogen bonds, they are more soluble in water and thus more likely to be undesirable constituents of industrial wastewater. In its most basic form, phenol is known as monohydroxy benzene (benzenol or, more simply, carbolic acid), and it was allegedly used as the first surgical antiseptic by British surgeon Joseph Lister in 1865.
Although phenols are now widely used as a disinfectant in cleaners and mouthwash (in low concentrations, of course), a small amount on the skin in concentrated form can cause severe burns and could eventually lead to death. Even at low concentrations, it is critical that Phenols be separated from wastewater as much as possible.
Phenol Sources in Wastewater
Because of their widespread use in household items, these pollutants are found in low concentrations in domestic wastewater. However, industrial plants such as pharmaceutical, agrochemical, and plastics and resin manufacturing facilities are also sources of phenol in wastewater. In these cases, the effluent must be managed at higher concentrations, necessitating the use of a specialized tertiary process.
Methods for Removing Phenol
Ozone (O3), activated carbon, or reverse osmosis can be used to treat phenol-contaminated effluent. However, ozone can be a complicated and'scattergun' treatment method, especially when the phenol is present in low concentrations. When compared to other companies, which localize the compound for efficient and targeted removal, the operational costs are lower.
Activated carbon can be labor intensive depending on the water quality because the process must be paused to change out or regenerate the carbon media.Water is passed through a membrane in reverse osmosis, and pollutants are 'filtered' out as a reject side-stream.
1: Activated Carbon for Phenol Removal
An activated carbon process can be used to manage this type of contamination. This method essentially absorbs the pollutant into the carbon's internal structure, resulting in a clean stream of water. Although phenol removal is achieved, the process must be paused and the carbon regenerated or replaced before the process can resume its efficiency. Once the carbon media has been'spent,' phenol removal by activated carbon is no longer possible. When carbon cannot be regenerated for reuse, it must be incinerated or disposed of in a landfill, both of which have negative environmental consequences.
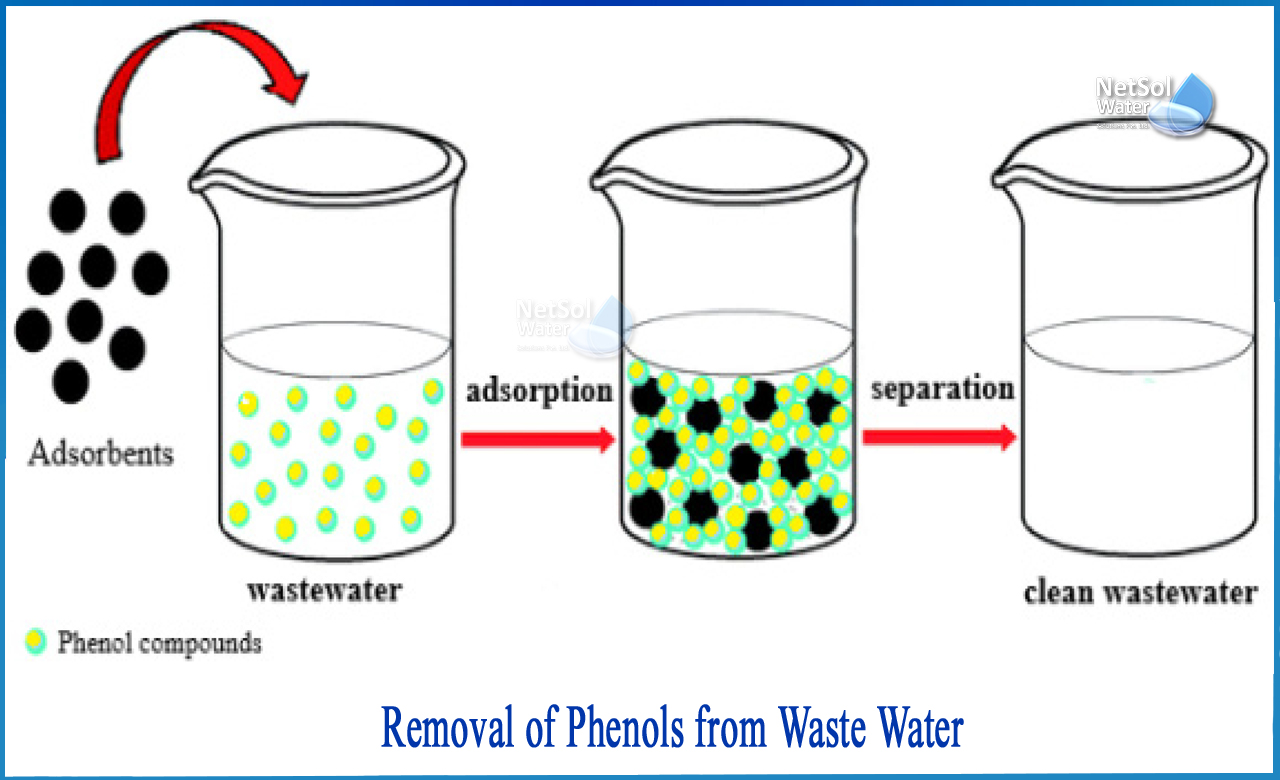
2: Adsorption and oxidation for phenol removal
One significant advantage of this system is that the adsorbent media used in this process is regenerated inside the system at the same time. In comparison to activated carbon, there are very few maintenance and operating costs to consider. In this case, phenol removal by adsorption is much more efficient and low-maintenance, with lower operational costs because the only input required is electricity to power the system.
3: Ozone of Phenol Removal
Although effective when done correctly, phenol removal by ozone is not always the most cost-effective option and necessitates complex chemistry tailored to each individual contaminant. Furthermore, the use of hydrogen peroxide (ozone AOP) can inhibit subsequent treatment steps, rendering this water unfit for reuse. Because there is no chemical dosing involved in the process, treated water is safe to reuse. It's also a simple 'plug and play' solution that doesn't require any complicated chemistry – just electricity.
Conclusion
Netsol Water can provide solutions to your water and waste water by providing various RO plants, Sewage treatment plants, effluent treatment plants and much more.
Netsol Water is Greater Noida-based leading water & wastewater treatment plant manufacturer. We are industry's most demanding company based on client review and work quality. We are known as best commercial RO plant manufacturers, industrial RO plant manufacturer, sewage treatment plant manufacturer, Water Softener Plant Manufacturers and effluent treatment plant manufacturers. Apart from this 24x7 customer support is our USP. Call on +91-9650608473, or write us at enquiry@netsolwater.com for any support, inquiry or product-purchase related query.