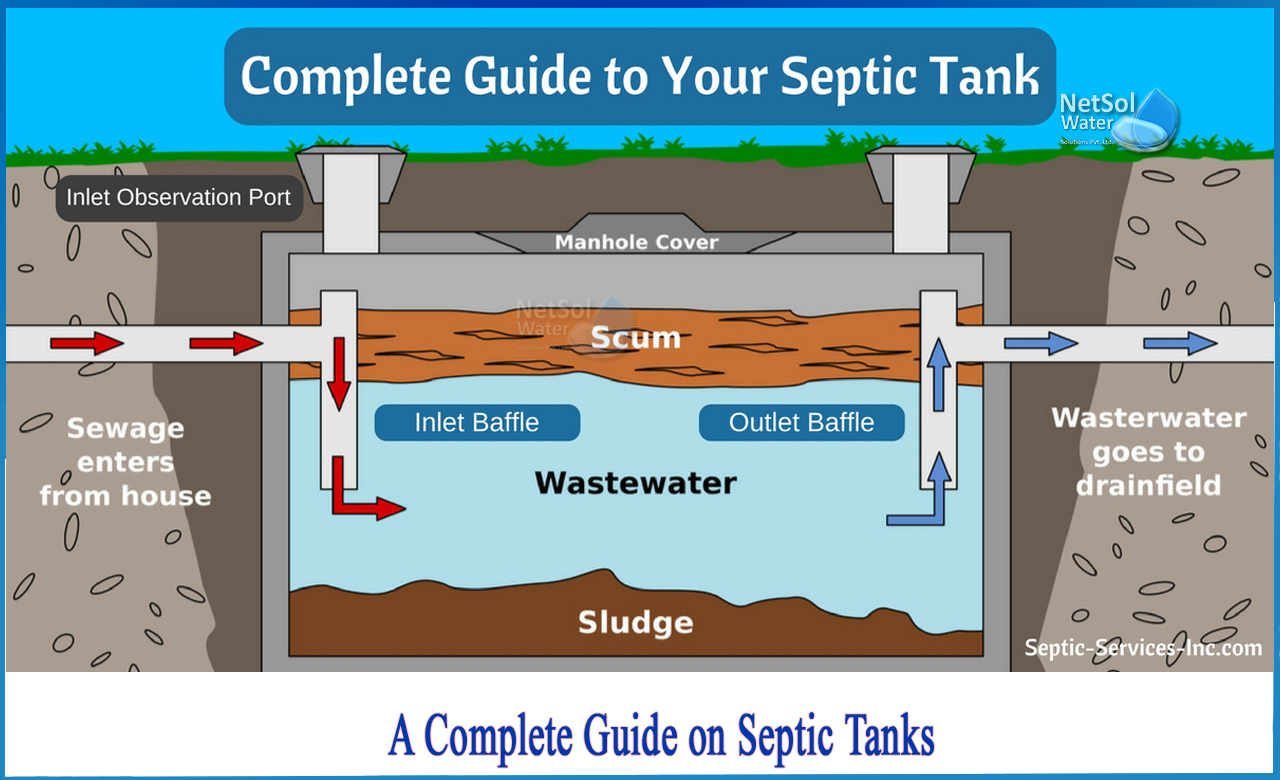
How to produce methane gas out of treated waste waters?
When organic waste decomposes in an oxygen-free environment, such as deep within a landfill, methane gas is released. Instead of being released into the environment, this methane may be trapped and utilised to generate electricity.
Sewage treatment systems begin by collecting solid sludge from wastewater. This sludge is then pre-treated with thermal hydrolysis in a sludge-to-energy system to optimise the quantity of methane it can create. The processed waste is then fed into an anaerobic digester, which breaks it down completely. The end result is a methane-rich gas, or biogas, which can be utilised for on-site energy or processed further and used in place of natural gas. Furthermore, the solid waste remains produce a nutrient-rich "digestate" that may be put to soil to enhance plant development.
Methane from waste water:
Through the anaerobic degradation of organic material, methane is released during the management and treatment of municipal wastewater. The majority of wealthy nations rely on to provide centralised aerobic wastewater treatment systems, municipal wastewater collection and treatment systems. These systems create modest amounts of methane emissions, but also huge volumes of methane emission levels of biosolids that can result in high methane rates emissions. In undeveloped countries where monitoring is limited or non-existent regarding wastewater treatment, the systems that do exist have a tendency to be anaerobic, resulting in higher methane emissions.
Wastewater's increasing importance:
In the next 40 years, the world's population is predicted to expand to more than 9 billion people, resulting in increased water demand and food consumption, as well as a corresponding increase in wastewater generation.Global methane emissions from wastewater are predicted to rise by about 19%.
Multiple advantages of methane capture and utilization:
Methane capture and use in wastewater treatment facilities offers several advantages:
• Lowers GHG emissions and related air pollutants.
• Offers a local source of energy, promoting energy independence.
• Turns a waste product into an income generator.
• Produces renewable energy that can be used to replace the consumption of fossil fuels.
• Creates jobs in the building and operation of projects.
• Improves the image of the local community as creative and sustainable.
Problems and solutions for methane extraction from wastewater:
Many sectors, including oil and gas refining, petrochemicals, and pharmaceuticals, use sophisticated processes that generate high-strength, difficult-to-treat wastewaters. These wastewaters contain at least one of the following hazardous elements: high amounts of biorefractory chemicals, poisonous compounds, halogenated organics, and aromatic or aliphatic hydrocarbons.Furthermore, their chemical oxygen demand (COD) levels can vary greatly, ranging from as little as 1,200 mg/L to more than 300,000 mg/L. Furthermore, certain process waters/wastewaters include significant quantities of salt, particularly chlorides, making cost-effective treatment particularly difficult.
Incineration or gasification are two existing treatment options for these high-salt wastewater streams. At 1,100°C (2,012°F), the former totally combusts the wastewater in the presence of extra oxygen, creating carbon dioxide, water, and salts. The latter uses stoichiometric oxygen to burn the wastewater, producing carbon monoxide and trace quantities of hydrogen. While these gases may be converted into more usable fuel gases, both processes are costly to operate and need backup units, which consume capital, operating expenditures, manpower, and space.Another treatment option for these high-salt wastewaters is wet air oxidation.
For more information, contact Netsol Water.
Netsol Water is Greater Noida-based leading water & wastewater treatment plant manufacturer. We are industry's most demanding company based on client review and work quality. We are known as best commercial RO plant manufacturers, industrial RO plant manufacturer, sewage treatment plant manufacturer, Water Softener Plant Manufacturers and effluent treatment plant manufacturers. Apart from this 24x7 customer support is our USP. Call on +91-9650608473, or write us at enquiry@netsolwater.com for any support, inquiry or product-purchase related query.