The most popular technique to determine the amount of organic matter in wastewater samples (i.e., relative strength) is called BOD. It is founded on the idea that aerobic biological decomposition, or, more specifically, the stabilisation of organic waste by microorganisms, will proceed as long as there is enough oxygen available.
Hence, BOD is a measure of the total amount of biodegradable organic compounds in the water. This also shows how much dissolved oxygen (milligrams per litre) is required over a specific period of time, for the biological breakdown of the elements of organic wastewater.
In this article, we will discuss about the calculation of BOD in sewage treatment plants with an example.
What is dissolved oxygen or DO?
Water usually contains some dissolved oxygen, which is referred to as DO. Aquatic life needs dissolved oxygen (DO) to survive, and it needs at least 4 PPM of dissolved oxygen to do so. When it comes to wastewater, organic materials must be broken down by this DO only.
What is Biological oxygen demand or biochemical oxygen demand or BOD?
Microorganisms such as aerobic bacteria break down organic materials in the presence of oxygen in sewage. The oxygen needed by aerobic bacteria to oxidise the biodegradable organic matter is referred to as BOD.
Significance of BOD in sewage treatment plants
1: It can be used to determine how much oxygen is necessary to stabilise the biodegradable organic material.
2: It also aids us in determining the sewage treatment plant's treatment unit size.
3: It gives us a sense of the procedure' effectiveness, or how much BOD is being eliminated. 4: In addition, we can discover sewage's strength.
BOD types
· First Stage BOD for carbonaceous matter;
· Second Stage BOD for nitrogenous matter;
How to calculate BOD for sewage treatment plants?
The "Dilution Technique" is the standard technique used in laboratories to determine a sewage sample's BOD. For BOD calculation in labs, 60% to 70% of the organic material is dissolved in 5 days. Also, 95% to 99% of the organic stuff is oxidised in 20 days.
The initial DO is discovered after diluting the water sample with aerated water, and it is then cultured for five days at 20 degrees Celsius. The DO, which is now referred to as the final DO, is found once more after these five days.
Thus,
BOD-5 at 20 °C = (Initial DO - Final DO) x Dilution Factor
The volume of the diluted sample divided by the volume of the undiluted sample, is known as the "dilution factor."
DF= Diluted Sample Volume/Undiluted Sample
How to derive the equation for BOD calculation?
It can be done by plotting the oxygen equivalent of organic matter present against time. The BOD is all that is left when calculating the "Oxygen Equivalent of Organic Matter Present."
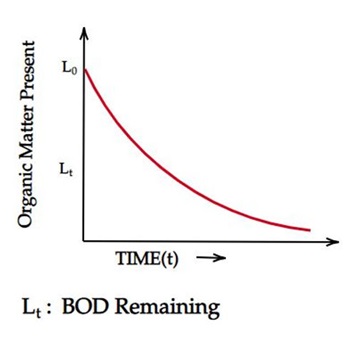
The rate of change of BOD directly varies with BOD remaining at that point in time (Lt)
So,
dLt/dt ∝ Lt
Integrating both the sides from interval Lo to Lt in time interval 0 to t,
Lt = Lo x e(-kt)
BOD Consumed,
Yt=Lo-Lt
At any time,
Yt = Lo - Lo x e(-kt)
Yt = Lo[1-e-kt]
To find out the solution in base “10” instead of base “e”, then,
t = Lo x 10-k't
Where k' = k/2.303
Here, the K' is also known as the de-oxygenation constant, which depends upon the temperature. The formulas are given at a standard temperature of 20°C. To convert to some other temperature,
K' (T Degree Cel.) = K' (20 Degree Cel.) x [1.047] ^(T-20)
To find the ultimate BOD, that happens when “t” tends to infinity;
Ultimate BOD = Lo
Factors on which BOD calculation depends
The calculation for BOD is based on the number of organisms present in the sewage sample, multiplied by their metabolic rate (metabolic rate is the speed at which they consume oxygen).
Therefore, to calculate the BOD of a sewage sample, multiply the number of organisms by their metabolic rate.
BOD = Number of Organisms x Metabolic rate
For example, if you have 2 million bacteria in a sample, and each consumes 0.04 mg of oxygen per hour, then the BOD would be:
BOD = 2,000,000 x 0.04 = 80,000 mg/L
This means that the BOD concentration is 80,000 mg/l.
Another example of BOD calculation
How is it possible for wastewater to have a BOD of 1,500 mg/L when pure water at 68°F has 9.1 mg/L of DO?
It is possible because of the serial dilution, which enables the step-by-step 10-fold concentration decrease of full-strength wastewater.
The final BOD result is just the difference between the original and final DO readings, multiplied by the dilution factor after dilution.
For instance, adding 1.0 mL of a wastewater sample at full concentration to 9.0 mL of DI water dilutes the wastewater by 0.1. The final BOD concentration must then be calculated, by multiplying the DO concentration (mg/L) reduction by 10.
Conclusion
The BOD test offers a reliable characterisation of wastewater/sewage when applied properly. Although, the 3 or 5 day wait time for the test (and perhaps 20 days) limits its effectiveness as a control tool, it has been anticipated to be a standard for regulatory bodies for many years.
Leading manufacturer of sewage treatment plants
Municipalities and industries can contact Netsol Water water treatment experts, for a variety of municipal sewage treatment services, including BOD removal from wastewater, COD removal from wastewater, and determining the optimal wastewater management strategy for their needs.
We have been providing wastewater treatment services to the industries and municipalities for many years. These services include water clarifying, sludge dewatering, chlorination and de-chlorination, phosphorus removal, odour control, etc. Call us at +91 9650608473 or email at enquiry@netsolwater.com for further information.



