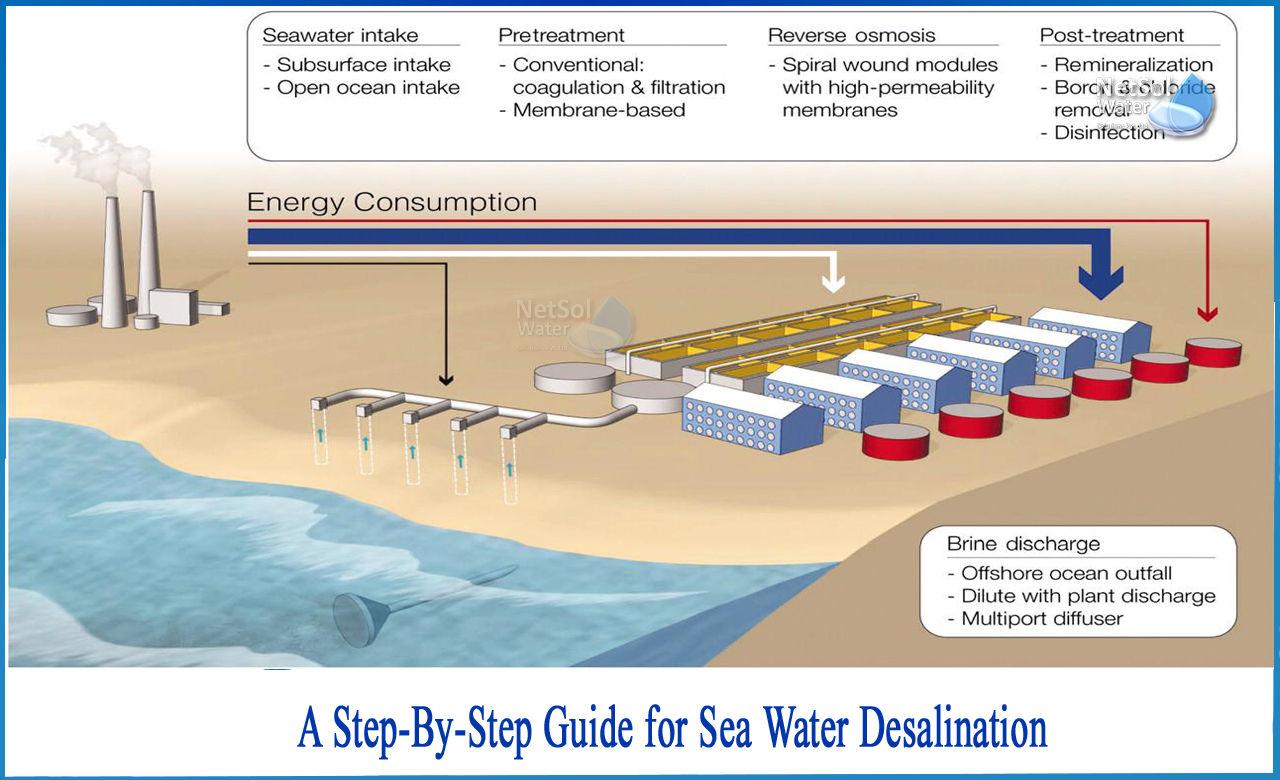
What is the process of desalination step by step?
Desalting is a separation process used to reduce the dissolved salt content of salt water to usable levels. All desalination processes include three liquid streams: salted water (brackish or seawater), low-salt produced water, and high-salt concentrated water (salt or rejected water).
Salty water comes from marine or underground springs. The desalination process separates it into two output streams (low salt production water and high salt concentrated stream). Some water-soluble substances, such as calcium carbonate, can be removed by chemical treatment, but other common components, such as sodium chloride, require a more sophisticated process, collectively called desalting. In the past, the difficulty and cost of removing various dissolved salts from water has made salt water an impractical source of drinking water.
The by-product of desalination
The by-product of desalination is salt water. Salt water is a concentrated salt solution (containing 35,000 mg/l or more dissolved solids) and should usually be disposed of by draining into a salt zone or surface water with high salinity. Salty water can also be diluted with treated wastewater and sprayed onto golf courses and other open spaces for disposal.
Technical Description1: Reverse osmosis (RO)
In the RO process, water flows through a permeable membrane to separate it from the molten salt from the pressurized saline solution. The remaining water supply passes through the pressure side of the reactor as brine. There is no heating or phase change. The main energy requirement is the initial pressurization of the water supply. The operating pressure range is 250-400 psi for brackish desalination and 800-1,000 psi for seawater desalination.
The water supply is pumped into a closed container against the membrane to apply pressure. As the resulting water passes through the membrane, the remaining water supply and brine solution are increasingly concentrated. A portion of this concentrated watering brine solution is removed from the container to reduce the concentration of the remaining dissolved salt. Without this introduction, the concentration of dissolved salts in the feed water would increase further, requiring higher energy inputs than ever to overcome the naturally increased osmotic pressure.
The reverse osmosis system consists of four major processes: (1) Pre-treatment, (2) Pressurization, (3) Membrane separation, and (4) post-treatment stabilization.
1: Pre-treatment: The water supply is compatible with the membrane by removing suspended solids, adjusting the pH, and adding a threshold suppressant to control the scale formation caused by components such as calcium sulphate.
2: Pressurization:The pressure is increased of the pre-treated feed water to an operating pressure suitable for the salt content of the membrane and feed water.
3: Separation: The application of water supply to the membrane assembly results in a stream of freshwater products and a stream of concentrated brine repulsion.
4: Stabilization: The water produced from the membrane assembly usually requires pH adjustment and degassing before entering the distribution system for use as drinking water. The product passes through an activation column where the pH value rises from about 5 to a value close to 7. Often, this water is drained into a storage tank for later use.
Conclusion
Netsol Water is a manufacturer of RO plants all over India. Even the reject water from the plants is re used for gardening and other processes.
Netsol Water is Greater Noida-based leading water & wastewater treatment plant manufacturer. We are industry's most demanding company based on client review and work quality. We are known as best commercial RO plant manufacturers, industrial RO plant manufacturer, sewage treatment plant manufacturer, Water Softener Plant Manufacturers and effluent treatment plant manufacturers. Apart from this 24x7 customer support is our USP. Call on +91-9650608473, or write us at enquiry@netsolwater.com for any support, inquiry or product-purchase related query.